Bayley Richardson1, Archana Ayyar2, Leigh Hickham3, Chinnadurai Mani4, Komaraiah Palle4, Mark Reedy1, 5*
1School of Medicine, Texas Tech Health Sciences Center, TX, USA
2Department of Obstetrics/Gynecology, Baylor College of Medicine, TX, USA
3School of Medicine, Louisiana State University Health Sciences Center, LA, USA
4Department of Cell Biology and Biochemistry, Texas Tech Health Sciences Center, TX, USA
5Department of Obstetrics/Gynecology, Texas Tech Health Sciences Center, TX, USA
*Corresponding Author: Mark Reedy, Associate Professor, Department Obestrics/Gynecology and Oncology, TTUHSC, Lubbock, TX, USA
Received: 26 May 2022; Accepted: 29 July 2022; Published: 22 August 2022
We describe a case of a 7-year-old female who presented to the emergency department (ED) with a three-day history of vaginal bleeding, progressive abdominal pain, and dehydration. Initial computerized tomography (CT) scan of the abdomen followed by a transabdominal ultra-sound of the pelvis identified bilateral, symmetric adnexal cysts and proliferative-phase endometrium with no evidence of inflammatory or malignancy-related findings. A gynecologic oncology consult led to surgical exploration diagnosing the child with an acute surgical abdomen. Surgery was performed through a vertical infra-umbilical, mini-laparotomy (4 cm) incision. Both intra-operative and final pathology confirmed bilateral ovarian, low malignant potential (LMP), granulosa cell tumors. Postoperative bone age was over 18 months less than the patient’s age. The discrepant bone age prompted investigation into pediatric syndromes characterized by juvenile hypothyroidism, isosexual precocious puberty, and granulosa cell tumors with delayed bone age. A rare diagnosis matching all criteria was identified as Van Wyk-Grumbach Syndrome (VW-GS). This syndrome was originally reported in 1960 by Drs. Van Wyk and Grumbach. They identified the discrepancies in bone age and how simple thyroid replacement results in complete resolution of this unique syndrome. Our literature review found few reports related to VWGS in the gynecologic oncology case studies. We hope this report will further assist in the appropriate diagnosis and management of similar cases to avoid unnecessary and invasive interventions and treatment.
Van Wyk-Grumbach Syndrome; Precocious Puberty; Hypothyroidism; Ovarian Cyst; Pediatric Syndrome
1. Introduction
In 1960, Drs. Van Wyk and Grumbach first published and “coined” this collection of related diagnoses, Van Wyck - Grumbach Syndrome (VW-GS) [1-3]. The initial report described a young female with precocious puberty, galactorrhea, ovarian cysts, and delayed bone age resulting from hypothyroidism. Since their initial description, the majority of literature appears in pediatric specialty journals [3-7]. Most gynecologic specialists would recommend surgical exploration to evaluate pediatric adnexal masses, while our pediatric colleagues likely would recommend medical management [8-10]. VW-GS appears to be most commonly caused by a non-congenital form of autoimmune hypothyroidism [11]. Due to the disruption in the negative feedback loop that thyroid hormone exerts on the hypothalamus and pituitary, there is an increase in TRH and TSH as observed in this patient (Table 1). The elevated hypothalamic-pituitary levels of TRH can suppress dopamine release [12]. Since pituitary dopamine prevents prolactin secretion, prolactin levels rise in severe hypothyroidism leading to other abnormalities such as galactorrhea [12]. Thyroid Stimulating Hormone, Follicle Stimulating Hormone (FSH), Luteinizing Hormone (LH), and beta Human Chorionic Gonadotropin (HCG) share a similar alpha-subunit, while their beta-subunit confers their specificity [13]. At normal physiologic levels, TSH is specific for its respective thyroid gland receptor. However, in severe hypothyroidism, especially with TSH >1000 mcg/ml, the TSH-alpha subunit can stimulate both FSH and LH receptors [14]. Simple thyroid hormone replacement ceases hyperstimulation of ovarian FSH and LH receptors, resolving the symptoms of VW-GS. In this article, we describe a prepubertal female that presented with classic Van-Wyk Grumbach symptomology. The aim of this report is to extend the knowledge in gynecologic oncology of a rare form of isosexual precocious pseudopuberty that is treated with conservative measures.
2. Case Presentation
A 7-year-old Hispanic female was brought to the ED complaining of lower abdominal pain and vaginal bleeding for several days. Her mother reported no fever or oral hydration (PK1) but worsening nausea without vomiting over the last 36 hours. Bowel function was unknown. Physical exam found the patient in obvious distress, lying in the fetal position and crying due to abdominal pain. Bowel sounds were absent. The abdomen was distended with guarding and rebound tenderness. The patient’s acute abdomen necessitated surgical evaluation. Prior to gynecologic oncology consultation, computerized tomography (CT) and abdominal ultrasound (AUS) identified bilateral 4 x 5 cm, simple ovarian cysts with thin-walled capsules. Other findings include a proliferative phase endometrium, absent ascites, normal appendix, and no obvious metastatic or inflammatory findings. Pre-operative laboratory tests found elevated tumor markers associated with ovarian granulosa cell tumors, i.e., elevated levels inhibin A and B, estradiol, and Mullerian inhibitory substance (MIS) and an elevated thyroid-stimulating hormone ((TSH), 1970 mlU/ml(normal 0.6-7.4 mIU/mL)), suggestive of severe hypothyroidism.
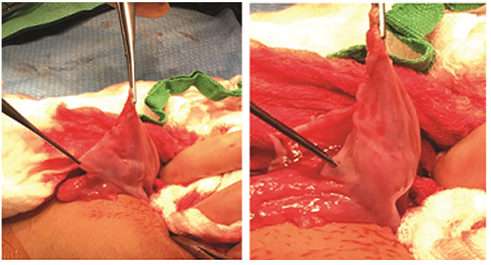
Following induction of general anesthesia, visual examination demonstrated Tanner stage 1 breast buds and absent axillary hair. The patient’s external genitalia were edematous without terminal hair or vaginal discharge. An obvious linea nigra was identified, resulting in an evaluation of serum progesterone, which was elevated as expected. Surgery was performed through a 4 cm infra-umbilical incision. No evidence of an acute inflammatory process was identified to explain the patient’s presentation. Bilateral ovarian cystectomies were performed by elevating each adnexa outside the abdomen, wrapping each with moist sponges, then hydro-dissecting the cysts from ovarian stroma using saline. Once the tissue planes were established, each cyst was dissected to prevent unexpected cyst fluid spill; a 22-gauge needle was used to aspirate both cysts and prevent accidental rupture. Each cyst was completely resected along with a piece of ovarian capsule attached for pathologic evaluation (Figure 1). Each ovarian cortex was pink, similar to tubal peritoneum, and void of follicles in either cortex. The frozen sections and final pathology reported bilateral, adult-type, granulosa cell neoplasm, low malignant potential (LMP). The bilateral ovarian GCT’s were identical in size, architecture, histology, IHC-staining and lacked the pathognomonic somatic FOXL2 mutation. Peritoneal cytology was negative. The postoperative course was uneventful, and the patient was discharged home three days later by the pediatric service.
Six-week post-operatively, the patient’s mother noticed vaginal bleeding. Unfortunately, follow-up appointments, scheduled labs tests, and medication compliance were not continued appropriately. Upon evaluation, the patient was found to have no signs/symptoms except vaginal bleeding. Lab evaluation test identified rising tumor markers (Inhibin A and B, estradiol). Abdominal ultrasound identified recurrent bilateral ovarian cysts (1-2 cm). Due to signs of progression, GnRH-agonist (Depot Lupron TM 7.5 mg IM) was administered (PK2) to block the production of sex hormones. Since levothyroxine was not started following hospital discharged, thyroid replacement was started at 25 micrograms daily and titrated every 4-6 weeks until normalized. To evaluate the patient’s hypophyseal-pituitary axis, CT was performed, and the results identified an enlarged pituitary gland. The pituitary gland was described as both homogenous and symmetric, consistent with pituitary gland hyperplasia. Further investigation identified the patient had a bone age of 18-month less than her age (7 years). The patient’s bone-age findings were contradictory to her elevated estradiol levels (326 pg/ml, normal <16pg/ml), which characteristically causes premature closure of the bony epiphyses and advanced bone-age.
With the list of findings highlighted by isosexual precocious puberty, the bilaterality, symmetric benign-appearing ovarian tumors, severity of the hypothyroidism, and a bone-age opposite of what was expected, our interest in pediatric syndromes ensued. Using a common search engine and the following characteristics - isosexual precocious puberty, bilateral ovarian tumors, hypothyroidism, and delayed bone age, a perfect match identified VW-GS. This syndrome appears to be a unique manifestation of severe, chronic hypothyroidism [15-19]. After reviewing the literature, especially the reports describing delayed bone age and how the syndrome resolves completely with thyroid replacement, the family was counseled on the importance of daily dosing of levothyroxine and follow-up. Soon after starting levothyroxine, the patient’s vaginal bleeding stopped, tumor markers decreased, and ovarian cysts resolved. Additionally, she had a rapid increase in height by 2-3 inches in 3-4 months. No further GnRH-agonist therapy was administered after the initial injection of levothyroxine. We cannot exclude the effects of a single goserelin (GnRH Agonist) injection, though the steady decline of all tumor markers such as the resolution of bilateral ovarian cysts, normalization of uterine architecture, and an increase in height and apparent lean muscle appeared after the thyroid replacement.
|
Laboratory tests |
Value |
Reference |
|
Prolactin |
140.9 |
0.3-12.9 ng/mL |
|
Progesterone |
4.4 |
< 2.0 ng/mL |
|
TSH |
1970 |
0.6-7.4 mIU/mL |
|
Free T4 |
0.1 |
0.8-1.5 ng/dL |
|
Estradiol |
326 |
<16 pg/mL |
|
Anti-Mullerian Hormone |
4.31 |
0.49-3.15 ng/mL |
|
17-hydroxyprogesterone |
264 |
< 90 ng/dL |
|
Inhibin A |
843 |
<98.0 pg/mL |
|
Inhibin B |
733 |
90 ng/dL or less |
|
Tumor marker AFP |
4.3 |
0.5-8.0 ng/mL |
|
Beta HCG |
<1.0 |
<10 mIU/mL |
Table 1: Pre-operative Laboratory values.
3. Discussion
On initial observation, the bilateral tumors in our patient resembled granulosa cell tumors. Adult-type granulosa cell tumors (AGTs) are considered a low-malignant potential neoplasm. A somatic mutation in the FOXL2 gene was found in 97-98% of all tumors [20]. This specific mutation is now considered a pathognomonic variant for AGTs. The more aggressive juvenile granulosa cell tumors lack the FOXL2 mutation. In our patient, both cystic ovarian tumors tested negative for the FOXL2 mutation. Although one would expect an advanced bone age due to elevated levels of estradiol in granulosa cell tumors, there is a striking difference in VW-G syndrome, a decreased bone age. The decreased bone age can be attributed to severe hypothyroidism, which delays the fusion of epiphysis [21]. Upon administering thyroid hormone replacement, the delayed bone age is rapidly corrected.
Van Wyk Grumbach syndrome is described as isosexual precocious psuedopuberty, multicystic enlarged ovaries, hypothyroidism, and delayed bone age. In addition, it is not uncommon to see enlarged pituitary glands [1, 22, 23]. This pituitary hyperplasia is secondary to hyperstimulation by TRH due to the loss of negative feedback from thyroid hormone [24]. Homogeneous pituitary hyperplasia has been reported in chronic, severe forms of hypothyroidism due to the demand to produce greater quantities of thyroid-stimulating hormone (TSH), as seen in our case [25]. When physiological equilibrium is restored by exogenous thyroid hormone, there is a regression of pituitary hyperplasia. With thyroid hormone playing a prominent role throughout the majority of the body, it is imperative to recognize and treat hypothyroidism in children in a timely manner. Typical symptoms include constipation, weight gain, lack of energy slow thinking, but in severe cases, hypothyroidism may cause restricted bone growth and delayed puberty [26]. The latter features are commonly associated with VW-G Syndrome. It should be of note that in this case, the 7-year-old 2nd grader was academically exceptional in math skills and had limited signs or symptoms despite her chronic, severe hypothyroidism. It is of interest to consider how this syndrome protects bone development from epiphyseal closure [27-30] and possibly, the brain from learning disabilities despite severe hypothyroidism during developmental years.
Although hypothyroidism is common in children, especially girls, which are four times more susceptible to hypothyroidism compared to boys, there are very few causes of VW-G syndrome [31]. Further, the molecular aspects or plausible genetic mutation of VW-G Syndrome have not yet been elucidated. Therefore, it is imperative we are aware of this syndrome to ensure early diagnosis and avoid unnecessary invasive procedures. This report contributes to the gynecologic oncology knowledge base by informing and educating our subspecialty about this rarely reported pediatric syndrome. Gynecologic oncologists with knowledge of Van Wyck Grumbach Syndrome should consider thyroid evaluation and bone-age determination to avoid misdiagnosis, unnecessary surgery or other therapies, and possible complications of these interventions [25].
Conflicts of Interest
The authors declare no conflict of interest.
Financial Support and Sponsorship
NIL
Acknowledgement
This work is partly supported by grants from Laura Bush Institute for Women’s Health and Weitlauf Endowment for Cancer Research
References
- Wyk JJV, Grumbach MM. Syndrome of precocious menstruation and galactorrhea in juvenile hypothyroidism: an example of hormonal overlap in pituitary feedback. The Journal of Pediatrics [Internet]. Elsevier 57 (1960): 416-435.
- Baranowski E, Högler W. An unusual presentation of acquired hypothyroidism: the Van Wyk-Grumbach syndrome. Eur J Endocrinol 166 (2012): 537-542.
- Christens A, Sevenants L, Toelen J, et al. Van Wyk and Grumbach syndrome: an unusual form of precocious puberty. Gynecol Endocrinol 30 (2014): 272-276.
- Rastogi A, Bhadada SK, Bhansali A. An unusual presentation of a usual disorder: Van Wyk-Grumbach syndrome. Indian J Endocrinol Metab 15 (2011): S141-S143.
- Razi SM, Gupta AK, Gupta DC, et al. Van Wyk-Grumbach Syndrome with Kocher-Debré-Sémélaigne Syndrome: Case Report of a Rare Association. Eur Thyroid J 6 (2017): 47-51.
- Oden Akman A, Tayfun M, Demirel F, et al. Association of Van Wyk Grumbach and Debre Semelaigne Syndromes with Severe Hypothyroidism. J Pediatr Adolesc Gynecol 28 (2015): e161-e163.
- Pant V, Baral S. Van Wyk Grumbach syndrome with precocious puberty and ovarian cysts: Value of thyroid function tests. Journal of Pediatric Surgery Case Reports (2019).
- Eskander RN, Bristow RE, Saenz NC, et al. A retrospective review of the effect of surgeon specialty on the management of 190 benign and malignant pediatric and adolescent adnexal masses. J Pediatr Adolesc Gynecol 24 (2011): 282-285.
- Luthra M, Kumar C. Surgical Management of Adnexal Masses in the Pediatric and Adolescent Age Group: Our Experience. J Indian Assoc Pediatr Surg 26 (2021): 287-293.
- Xac MC, Jetelina KK, Jarin J, et al. Benign, Borderline, and Malignant Pediatric Adnexal Masses: A 10-Year Review. J Pediatr Adolesc Gynecol 34 (2021): 454-461.
- Jagadhish TK. Van Wyk and Grumbach Syndrome (A Syndrome of Incomplete Isosexual Precocity and Juvenile Hypothyroidism). Med J Armed Forces India 58 (2002): 343-345.
- Mariotti S, Beck-Peccoz P. Physiology of the Hypothalamic-Pituitary-Thyroid Axis. In: Feingold KR, Anawalt B, Boyce A, Chrousos G, de Herder WW, Dhatariya K, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc. (2000).
- Styne DM, Grumbach MM. CHAPTER 25 - Puberty: Ontogeny, Neuroendocrinology, Physiology, and Disorders. In: Melmed S, Polonsky KS, Larsen PR, Kronenberg HM, editors. Williams Textbook of Endocrinology (Twelfth Edition) [Internet]. Philadelphia: W.B. Saunders (2011): 1054-1201.
- Anasti JN, Flack MR, Froehlich J, et al. A potential novel mechanism for precocious puberty in juvenile hypothyroidism. J Clin Endocrinol Metab 80 (1995): 276-279.
- Shivaprasad KS, Dutta D, Jain R, et al. Huge bilateral ovarian cysts in adulthood as the presenting feature of Van Wyk Grumbach syndrome due to chronic uncontrolled juvenile hypothyroidism. Indian J Endocrinol Metab 17 (2013): S164-S166.
- Zhu W, Yang Y, Zhao Y, et al. Primary juvenile hypothyroidism concurrent with huge bilateral cystic ovaries: Case report and literature review. Arch Pediatr 28 (2021): 96-100.
- Guerrero N, Athanassaki ID, Sebastian MR. Prolonged Untreated Disease and Limited English Proficiency: A Case of Van Wyk-Grumbach Syndrome. J Adolesc Health 69 (2021): 171-174.
- Lee S-J, Moon J-E, Lee G-M, et al. An Alport syndrome boy with Van Wyk-Grumbach syndrome induced by prolonged untreated congenital hypothyroidism. Ann Pediatr Endocrinol Metab 25 (2020): 132-136.
- Reddy P, Tiwari K, Kulkarni A, et al. Van Wyk Grumbach Syndrome: A Rare Consequence of Hypothyroidism. Indian J Pediatr 85 (2018): 1028-1030.
- Köbel M, Gilks CB, Huntsman DG. Adult-type granulosa cell tumors and FOXL2 mutation. Cancer Res 69 (2009): 9160-9162.
- Reed BG, Carr BR. The Normal Menstrual Cycle and the Control of Ovulation. In: Feingold KR, Anawalt B, Boyce A, Chrousos G, de Herder WW, Dhatariya K, et al., editors. Endotext [Internet]. South Dartmouth (MA): MDText.com, Inc. (2000)
- Hunold A, Alzen G, Wudy SA, et al. Ovarian tumor in a 12-year old female with severe hypothyroidism: A case of Van Wyk and Grumbach syndrome. Pediatr Blood Cancer 52 (2009): 677-679.
- Durbin KL, Diaz-Montes T, Loveless MB. Van wyk and grumbach syndrome: an unusual case and review of the literature. J Pediatr Adolesc Gynecol 24 (2011): e93-e96.
- Al-Gahtany M, Horvath E, Kovacs K. Pituitary hyperplasia. Hormones (Athens) 2 (2003): 149-158.
- De Sousa SMC, Earls P, McCormack AI. Pituitary hyperplasia: case series and literature review of an under-recognised and heterogeneous condition. Endocrinol Diabetes Metab Case Rep 2015 (2015): 150017.
- Bauer AJ, Wassner AJ. Thyroid hormone therapy in congenital hypothyroidism and pediatric hypothyroidism. Endocrine 66 (2019): 51-62.
- Egodawaththe NS, Seneviratne SN, Gunasekara S, et al. Van Wyk-Grumbach syndrome and oligosyndactyly in a 6-year-old girl: a case report. J Med Case Rep 14(2020): 166.
- Gomez GA, Aghajanian P, Pourteymoor S, et al. Differences in pathways contributing to thyroid hormone effects on postnatal cartilage calcification versus secondary ossification center development. Elife 11 (2022): e76730.
- Aghajanian P, Xing W, Cheng S, et al. Epiphyseal bone formation occurs via thyroid hormone regulation of chondrocyte to osteoblast transdifferentiation. Sci Rep 7 (2017): 10432.
- Williams GR, Bassett JHD. Thyroid diseases and bone health. J Endocrinol Invest 41 (2018): 99-109.
- 5 Surprising Facts about “Low Thyroid” in Children and Teens (2022).