Azka Ali1*, Sangeetha Krishnamoorthy2, Gloria John Reng3, Behram Khan Ghazi4, Qingqing Wen5, Sheheryar Sharif6, Anam Hamid7, Asma Nasir7, Muhammad Humza Qamar6, Pratiksha Pramodrao Kapartiwar8, Sultana Rajia9, Zoia Akram10, Muhammad Arslan Aslam11, Jennifer Alicea Maldonado12, Vrinda Goel13, Annerin Maxwell14
Review Article
1King Edward Medical University, Lohore, Pakistan
2Tianjin Medical University, Tianjin, China
3V.N Karazin Kharkiv National Medical University, Kharkiv Oblast, Ukraine
4Faisalabad Medical University/Punjab Medical College, Punjab, Pakistan
5University of California, Los Angeles/Fielding School of Public Health, California, USA
6Frontier Medical and Dental College, Khyber Pakhtunkhwa, Pakistan
7Dow University of Health and Sciences, Sindh, Pakistan
8Shree Vasantrao Nike Government Medical College, Maharashtra, India
9Sher E Bangla Medical College, Barishal, Bangladesh
10Allama Iqbal Medical College, Punjab, Pakistan
11Nishtar Medical University, Punjab, Pakistan
12Universidad Autónoma de Guadalajara, Zapopan, Mexico
13Jawaharlal Nehru Medical College, Karnataka, India
14American University of Antigua, St. John’s, Osbourn, Antigua & Barbuda
*Corresponding author: Azka Ali, Department of Internal Medicine, King Edward Medical University, Lohore, Pakistan
Received: 19 April 2021; Accepted: 28 April 2021; Published: 04 May 2021
Coronavirus disease-2019 (COVID-19) is a global medical burden. Severe acute respiratory coronavirus 2 (SARS-CoV-2) can cause significant mortality and morbidity by implicating pulmonary infection and respiratory failure. It has a corresponding inflammatory impact on various organ systems as well. A considerable drop in the number of patients attending the emergency department, undergoing elective procedures and surgeries has been reported in several studies from different countries affected by the pandemic. In the setting of the ongoing pandemic, cardiologic emergency of the acute coronary syndrome (ACS) has raised several critical clinical issues regarding its incidence, lower hospital admission rates, management, and therapeutic uncertainties. It is primarily due to the risk of association between respiratory infections and Covid-19 induced myocardial injury, hesitation by the patients to visit the high-risk hospital environment, and missed diagnoses. Consequently, short and long-term ACS complications are expected in the near future. Our study sought to investigate the correlation between diagnosis and management of ACS and pathophysiological links with SARS-CoV-2 infection, the decline in ACS cases across the world in the time of the pandemic, with a focus on the prediction of upcoming complications and also modifying the social and clinical approach towards ACS among the health care personnel to prevent the serious complications in acute cardiology care.
Covid-19, Acute Coronary Syndrome
1. Introduction
Since its outbreak in Wuhan, China in December 2019, the severe acute respiratory syndrome (SARCoV-2), a highly infectious agent causing the clinical syndrome of Coronavirus disease-2019 (COVID-19), evolved into the ongoing global pandemic affecting the healthcare system all around the world [1, 2]. In early March 2020, the World health organization (WHO) announced it a public health emergency on the international level with the total number of confirmed cases 126,890,643 as of March 29, 2021, including 2,778,619 confirmed deaths [3]. The constellation of symptoms caused by COVID-19 includes fever, cough, myalgia, and shortness of breath within 2-14 days of patient exposure to this virus which can then progress to covid-19 related pneumonia and develop to acute respiratory distress syndrome (ARDS) in one-third of the patients, one of the most formidable complications of this disease requiring intensive care unit (ICU) admission [4]. Although the characteristic clinical SRS-CoV-2 manifestations prevail in the respiratory system, it can cause multiple organ dysfunction through a systemic inflammatory response. Recent preliminary data demonstrate that severe cardiovascular damage can occur through COVID-19 induced myocardial injury and have a fatal outcome in patients with underlying cardiac diseases or pro-inflammatory cardiovascular risk factors [5]. However, observations in cardiology settings raised concern about the incidence, presentation, and management of ACS. Multiple international consensus and researches have been going on to identify and validate useful predictive factors to help address the developing potentially serious complications related to ACS management during the pandemic.
Acute coronary syndrome accounts for 1 million hospital admissions annually in the US and is still considered a substantial cause of morbidity and mortality, with almost 1.8 million yearly deaths attributed to coronary heart disease (CHD) in people older than 35 years [6] and 20% of all deaths in Europe [7]. A recent report from the 2016 Heart Disease and Stroke Statistics update of the American Heart Association (AHA) showed that 15.5 million people ≥20 years of age have CHD in the USA [8]. The recent viral pandemic related to SARS-CoV-2 infection has drastically collapsed the health care system across various countries due to overwhelming patient load, depleting the healthcare resources and posing a new challenge to the modern health care system [9]. It has led to the deferral of screening and elective procedures for coronary artery disease (CAD) and has revolutionized the routine clinical practices in ACS management. Moreover, with national coronavirus lockdowns, a decline in the incidence of ACS cases has been reported from around the globe [10] due to lower patients referral to the emergency department, lack of timely diagnosis, and management of concurrent diseases that share overlapping symptoms between non-COVID ACS and COVID-19 induced myocardial injury (COVID-AMI), viral myocarditis, and stress-induced cardiomyopathy [11]. In the context of delayed ACS management and lack of standardized protocols during the pandemic, the increased emergence of short-term and long-term MI complications are foreseen, which will probably support the need for continuous innovation and modernization in public health resources and the healthcare system [12].
In the present discussion, we included relevant published literature, case reports and retrospective series to show that a response-adapted approach may effectively guide therapeutic decisions in-terms of treatment intensifications, without compromising the protection of health care personnel and also recommended guidelines towards the management of ACS in acute cardiology setting during the ongoing pandemic.
2. Acute Coronary Syndrome Pathogenesis and Diagnosis with ARTI
The term “Acute Coronary Syndrome” encompasses a cluster of coronary heart disease events, including unstable angina (UA), ST-segment elevation acute myocardial ischemia (STEMI), and non-ST segment elevation myocardial infarction (NSTEMI) due to various thrombotic coronary artery etiologies (usually atherosclerosis), reducing/occluding the blood flow towards the heart [13]. Moreover, the elevation of cardiac troponin (cTn) above 99th percentile of upper reference limit (URL) along with a clinical spectrum of cardiac ischemia in the setting of evidence of electrocardiogram (ECG) changes, angiographic findings, and imaging evidence are required to assist in stratifying the patient’s risk of having the acute coronary syndrome, as described by the fourth universal definition of myocardial infarction [14]. The serum cardiac enzymes troponin T and I (cTI) are generally considered the most sensitive biomarkers for the determination of myocardial injury, although early serum cardiac markers including myoglobin and creatine kinase-MB subforms/isoforms, are also used worldwide [15].
The clinical manifestations of type 2 MI include atypical signs and symptoms such as dyspnea [16], making its diagnosis challenging and causing considerable confusion in differentiating it from non-cardiac diseases or comorbidities with overlapping features that may mask its presentation [17]. The phenomenon of myocardial necrosis or type 2 MI can be induced by disruption between oxygen supply and demand, provoked by different clinical etiologies such as hypoxemia in conjunction with respiratory failure and infectious diseases (predominantly sepsis) [18, 19]. The risk of myocardial ischemia is 17 folds higher in patients with acute respiratory infections, mainly due to disruption in ventilation-perfusion ratios, a functional consequence of underlying hypoxemia, and loss of hypoxic vaso-constrictive reflex. Reduced oxygen supply then activates the sympathetic nervous system, increasing heart rate and cardiac contractility and putting stress on myocardial oxygen demand leading to myocardial infarction [20]. It has also been suggested that pro-inflammatory states triggered by respiratory tract infections along with pronounced components of micro-vascular thrombosis are implicated in the immuno-pathogenesis of ACS [21]. Moreover, respiratory infections such as those caused by the influenza virus and other respiratory viruses have been associated with an increased risk of MI due to susceptible gene expression that stimulates platelet activation. Hence, correlating with hyper-coagulation and thrombosis, and contributing to increased risk of MI [18, 22].
The hyper inflammatory response under the influence of circulating cytokines, Interleukin (IL-1, IL-6, and IL-8), and tumor necrosis factor-alpha (TNF-α) generated at the infection site stimulates the release of macrophages and T-cells inside the atherosclerotic plaques. These cells activate matrix metalloproteinase and peptidase, which accompanied by increased vascular permeability promote fibrous cap formation [23]. As a result of intraplaque inflammatory activity, degradation of extracellular matrix and oxidative burst occurs, releasing phospholipids, tissue factor, collagen and platelet-adhesive matrix elements which contribute to plaque instability and thrombus formation, typical pathogenesis of type 1 MI [24]. Furthermore, pro-thrombotic states provoked by cytokines lead to fibrin deposition with subsequent tissue damage and the development of microangiopathic pathology in multiple organs, facilitating coronary artery thrombosis at plaque disruption site. The diagnosis of myocardial infarction may go unnoticed in critically ill patients [25] with acute respiratory failure, as evident by postpartum studies which showed the prevalence of undiagnosed acute myocardial infarction (AMI) ranging from 5% to 25% in patients who died from acute respiratory failure [26].
3. COVID-19 Triggered AMI
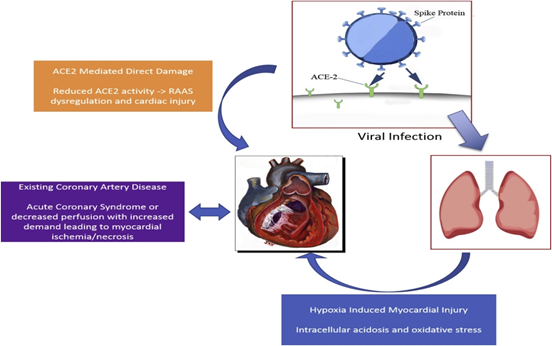
The prevalence of acute myocardial injury was first reported as a major complication in 12% of 41 hospitalized SARCoV-2 infected patients by Huang et al. [4]. A cluster of other studies demonstrated the acute cardiac injury in SARS-CoV-2 infected patients, and acute cardiac injury is considered to pose a higher mortality risk in COVID-19 patients with underlying cardiovascular morbidities and in patients who had a severe clinical presentation [27]. The suggested possible mechanisms by which SARS-CoV-2 triggers acute myocardial infarction and cardiac muscle injury are multifactorial (Figure 1). However, the definitive link between AMI and SARS-CoV-2 is yet to be confirmed from a large cohort of patients worldwide. Acute myocardial injury is typically documented in the advanced stages of the disease and is associated with a worse prognosis. Wang et al. found 7.2% of patients had acute myocardial dysfunction in 138 hospitalized patients with Covid-19 and 22.2% of them were associated with ICU admissions [28]. Li et al. reported acute myocardial injury in at least 8% of the patients with 13 folds higher risk in patients with severe clinical presentation admitted to the critical care unit [29]. Moreover, data from the National Health Commission of China reported that 11.8% of the COVID-19 patients who died without underlying cardiovascular diseases had significant heart damage, as evident from elevated levels of cardiac troponin I (cTnI) or in-hospital cardiac arrest [30, 31].
COVID-19, through its spike (S) protein, binds to the angiotensin-converting enzyme (ACE-2) receptor, expressed abundantly in heart and lungs epithelium. The endothelial ACE through angiotensin II receptor type 1 (AT-1) then converts angiotensin 1 to angiotensin 2 which heightens the accumulation of inflammatory cells in the endothelium, rendering to endothelial inflammation/injury, vasoconstriction, pro-inflammatory and pro-oxidative state [32]. Even though the relationship of acute myocardial injury with SARS-CoV-2 is not yet clear, the hyper inflammation and systemic inflammatory response in SARCoV-2 could be the primary trigger leading to acute myocardial injury or myocarditis [33]. Hypoxemia resulting from the violation of ventilation-perfusion mismatch may potentiate the cardiac muscle damage [34]. Hypercoagulable state and development of microangiopathic pathology [35] accompanied by protective signaling pathway due to down regulation of ACE-2 receptors with the pronounced component of microvascular coronary artery thrombosis can contribute to myocardial dysfunction [32]. Furthermore, the specific damage caused by SARS-CoV-2 could be due to direct invasion of cardiac myocytes, probably via ACE -2 receptor on cardiac endothelium as evident by the presence of SARS-CoV-2 RNA in 35% of the autopsied hearts in patients who died during COVID outbreak [36].
Although the proposed mechanism of action for COVID-AMI resembles other respiratory virus infections, cytokine storm is conferred to be responsible more for AMI than isolated cardiac injury as confirmed from the elevation of cardiac troponin (cTn) in association with other inflammatory biomarkers such as interleukin-6, D-dimer, lactate dehydrogenase (LDH) [37]. Moreover, differential diagnoses of COVID-AMI which can present with overlapping symptoms, EKG changes, and wall motion abnormalities such as viral myocarditis and cardiomyopathy become a challenge for clinicians to reach a definitive diagnosis due to underutilization of coronary angiography and other imaging modalities during times of pandemic.
4. Prevalence of ACS during COVID-19 Pandemic
Reports from all over the world demonstrate the astonishing decline in ACS cases and increase in the incidence of delayed presentation of MI with symptoms of chronic heart failure and sudden cardiac arrest due to the relevant impact of COVID-19 pandemic together with the risk of early morbidity and mortality [38]. COVID-19 has impacted inpatient cardiology capacity due to the transformation of many wards into COVID-19 reserved units [39]. Also, ACS admissions have declined significantly as hospitals are perceived as possible sites for COVID exposure. Prof. B. Casadei, working at Oxford University, stated that ACS admissions decreased by 75% in areas hardly hit by COVID. In Italy, a reduction in hospitalization for STEMI (26.5%) and NSTEMI (65.1%) has been reported. Similar reports from the USA and Spain document the downfall of 38% and 40% in cardiac catheterization laboratory activation for STEMI [40, 41]. A retrospective analysis from northern Italy evaluated the incidence rate of ACS hospitalization from 15 hospitals and found the admission rate to be drastically decreased to 13.3 admissions per day from February 20, 2020 to March 31, 2020 as compared to the previous year (18.9 admissions per day with IRR of 0.70; p<0.001) and to admissions earlier in the pandemic 18.0 admissions per day (IRR 0.74 ; p<0.001) [11]. Metzler et al. documented the decline of 39.4% for ACS-associated hospital admissions in Austria in a retrospective survey conducted on consecutive patients [10].
A recent study by Trabattoni et al. from Lombardy, Italy showed the increased rate of in-hospital mortality due to substantial delayed (>24hr) presentation of STEMI (41% cases) in 2020 in comparison to 20% in the 2019 year, 38% versus 10% respectively [42]. A similar important aspect is described by Tam et al. who compared a reduction in hospitalization and late presenting MI than previous years during the study period of January 25, 2020 to February 10, 2020 owing to patients’ hesitation to go to the hospitals due to stay-at-home orders and apprehension of contracting in-hospital infections [38]. A 4.97 increased incidence of out of hospital sudden cardiac arrest was reported in New York City during the surge of pandemic in comparison to the previous year during the same time period [43]. Similar reports from Italy are documented by Baldi et al., demonstrating higher incidence of out-of-hospital sudden cardiac arrest during outbreak of COVID-19 infection [44]. The Canadian Association of Interventional Cardiology (CAIC) announced the formation of the North American Covid-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI) on April, 2020, to document the important information about this topic that might prove helpful in timely and appropriate decision making strategies regarding treatment and management of ACS cases [45].
5. Reperfusion Strategies in Critically Ill Patients
Coronary heart disease is still a major cause of mortality worldwide. One of the most formidable global challenges to the healthcare settings during the pandemic is finding the balance between the risks of ACS treatment and SARS-CoV-2 infection control [46]. Drug therapy for MI should be administered cautiously due to possible interaction between anti-platelets (clopidogrel and ticagrelor) and antivirals particularly lopinavir–ritonavir [47]. It has been seen that low dose aspirin and other NSAIDs can be used safely as antiplatelet agents in Covid-19 patients [48]. Beta blockers, statins, angiotensin receptor blockers (ACE- inhibitors), and angiotensin-II receptor blockers (ARBs) should also be ensured to palliate symptoms and reduce long term mortality risk [49]. However, the European Association of Percutaneous Cardiovascular Interventions (EAPCI) and the Acute Cardiovascular Care Association (ACVC) published latest recommendations that invasive therapy should not be delayed during COVID-19 pandemic and that the standard therapy of primary percutaneous coronary intervention (PCI) should remain the first therapeutic option for ACS management if administered within given timeframe in compliance with preventive therapies for healthcare workers [50].
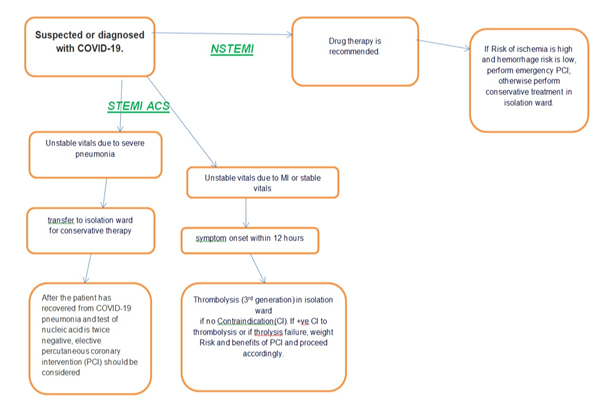
The Society for Cardiovascular Angiography and Interventions (SCAI), ACC and the American College of Emergency Physicians (ACEP) stated ACS management guidelines during COVID-19 pandemic in the USA. It was proposed that the initial fibrinolysis should be preferred within 30 minutes of STEMI diagnosis for the COVID-19 patients who cannot get early reperfusion therapy with eventual transfer to appropriate PCI Hub centers if needed. However, thrombolysis should be limited to specific cases and must not be considered a standard of care strategy for AMI management to avoid potential harmful adverse events related to its use [51]. It has been postulated that immediate PCI should not be done in severely ill COVID patients with STEMI or high-risk ACS. Guidelines by two hospitals in China are as follows for STEMI patients: Thrombolysis is preferred over PCI in COVID suspected/confirmed patients. For NSTEMI-UA, the first thing to be done is to rule out COVID-19 infection. Daniels et al. further approved these guidelines as elaborated in the following algorithm [52]. Nevertheless, very high-risk STEMI should be treated similar to STEMI management without waiting for screening results.
6. Conclusion
In the midst of the widespread coronavirus disease 2019 (COVID-19) pandemic with substantial disease burden across the globe, unrecognized ACS cases and reduction in the AMI hospitalization have raised a recent concern among the health care providers. Our study highlights the therapeutic challenges in ACS diagnosis and management during the pandemic. Further prospective international studies are warranted to differentiate the potential mechanistic link between primary ACS and cardiac damage caused by COVID-19. International global registry and randomized trials from a global perspective can help develop a standardized approach in ACS diagnosis and treatment to optimize the early morbidity and mortality along with documentation of actual prevalence of ACS. A global response to establish a primary PCI center to launch a timely 24/7 PCI-reperfusion therapy, based on staff protection, appropriate PPE, and Catheterization-lab availability is imperative.
References
- Livingston E, Bucher K. Coronavirus Disease 2019 (COVID-19) in Italy. Jama 323 (14): 1335.
- CENTER JHUaMCR. COVID-19 DATA IN MOTION (2021).
- Organization WH. WHO Coronavirus (COVID-19) Dashboard (2021).
- Huang C, Wang Y, Li X, et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet (London, England) 395 (2020): 497-506.
- Shi S, Qin M, Shen B, et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA cardiology 5 (2020): 802-810.
- Eisen A, Giugliano RP, Braunwald E. Updates on Acute Coronary Syndrome: A Review.JAMA cardiology 1 (2016): 718-730.
- Townsend N, Wilson L, Bhatnagar P, et al. Cardiovascular disease in Europe: epidemiological update 2016. European heart journal 37 (2016): 3232-3245.
- Mozaffarian D, Benjamin EJ, Go AS, et al. Heart disease and stroke statistics—2016 update: a report from the American Heart Association. circulation 133 (2016): e38-e360.
- Remuzzi A, Remuzzi G. COVID-19 and Italy: what next?. Lancet (London, England) 395 (2020): 1225-1228.
- Metzler B, Siostrzonek P, Binder RK, et al. Decline of acute coronary syndrome admissions in Austria since the outbreak of COVID-19: the pandemic response causes cardiac collateral damage. European heart journal 41 (2020): 1852-1853.
- De Filippo O, D’Ascenzo F, Angelini F, et al. Reduced rate of hospital admissions for ACS during Covid-19 outbreak in Northern Italy. New England Journal of Medicine 383 (2020): 88-89.
- De Rosa S, Spaccarotella C, Basso C, et al. Reduction of hospitalizations for myocardial infarction in Italy in the COVID-19 era. European heart journal 41 (2020): 2083-2088.
- Amsterdam EA, Wenger NK, Brindis RG, et al. 2014 AHA/ACC guideline for the management of patients with non-ST-elevation acute coronary syndromes: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines. Circulation 130 (2014): e344-e426.
- Thygesen K, Alpert JS, Jaffe AS, et al. Fourth universal definition of myocardial infarction (2018). European heart journal 40 (2019): 237-269.
- Achar SA, Kundu S, Norcross WA. Diagnosis of acute coronary syndrome. American family physician 72 (2005): 119-126.
- Lippi G, Sanchis-Gomar F, Cervellin G. Chest pain, dyspnea and other symptoms in patients with type 1 and 2 myocardial infarction. A literature review. International journal of cardiology 215 (2016): 20-22.
- Stein GY, Herscovici G, Korenfeld R, et al. Type-II myocardial infarction--patient characteristics, management and outcomes. PloS one 9 (2014): e84285.
- Musher DM, Abers MS, Corrales-Medina VF. Acute Infection and Myocardial Infarction. The New England journal of medicine 380 (2019): 171-176.
- Sandoval Y, Smith SW, Sexter A, et al. Use of objective evidence of myocardial ischemia to facilitate the diagnostic and prognostic distinction between type 2 myocardial infarction and myocardial injury. European heart journal Acute cardiovascular care 9 (2020): 62-69.
- Ruane L, Buckley T, Hoo SYS, et al. Triggering of acute myocardial infarction by respiratory infection. Internal medicine journal 47 (2017): 522-529.
- Davidson JA, Warren-Gash C. Cardiovascular complications of acute respiratory infections: current research and future directions. Expert review of anti-infective therapy 17 (2019): 939-942.
- Rose JJ, Voora D, Cyr DD, et al. Gene Expression Profiles Link Respiratory Viral Infection, Platelet Response to Aspirin, and Acute Myocardial Infarction. PloS one 10 (2015): e0132259.
- Crea F, Liuzzo G. Pathogenesis of acute coronary syndromes. Journal of the American College of Cardiology 61 (2013): 1-11.
- Fuster V, Badimon L, Badimon JJ, et al. The pathogenesis of coronary artery disease and the acute coronary syndromes (1). The New England journal of medicine 326 (1992): 242-250.
- Arlati S, Brenna S, Prencipe L, et al. Myocardial necrosis in ICU patients with acute non-cardiac disease: a prospective study. Intensive care medicine 26 (2000): 31-37.
- Soeiro Ade M, Ruppert AD, Canzian M, et al. Postmortem diagnosis of acute myocardial infarction in patients with acute respiratory failure: demographics, etiologic and pulmonary histologic analysis. Clinics (Sao Paulo, Brazil) 67 (2012): 213-217.
- Zhou F, Yu T, Du R, et al. Clinical course and risk factors for mortality of adult inpatients with COVID-19 in Wuhan, China: a retrospective cohort study. The lancet 395 (2020): 1054-1062.
- Wang D, Hu B, Hu C, et al. Clinical Characteristics of 138 Hospitalized Patients With 2019 Novel Coronavirus-Infected Pneumonia in Wuhan, China. Jama 323 (2020): 1061-1069.
- Li B, Yang J, Zhao F, et al. Prevalence and impact of cardiovascular metabolic diseases on COVID-19 in China. Clinical research in cardiology : official journal of the German Cardiac Society 109 (2020): 531-538.
- Zheng YY, Ma YT, Zhang JY, et al. COVID-19 and the cardiovascular system. Nature reviews Cardiology 17 (2020): 259-260.
- Lippi G, Lavie CJ, Sanchis-Gomar F. Cardiac troponin I in patients with coronavirus disease 2019 (COVID-19): Evidence from a meta-analysis. Progress in cardiovascular diseases 63 (2020): 390-391.
- Oudit GY, Kassiri Z, Jiang C, et al. SARS-coronavirus modulation of myocardial ACE2 expression and inflammation in patients with SARS. European journal of clinical investigation 39 (2009): 618-625.
- Zhou Y, Fu B, Zheng X, et al. Pathogenic T cells and inflammatory monocytes incite inflammatory storm in severe COVID-19 patients. National Science Review (2020).
- Kubasiak LA, Hernandez OM, Bishopric NH, et al. Hypoxia and acidosis activate cardiac myocyte death through the Bcl-2 family protein BNIP3. Proceedings of the National Academy of Sciences of the United States of America 99 (2002): 12825-12830.
- Han H, Yang L, Liu R, et al. Prominent changes in blood coagulation of patients with SARS-CoV-2 infection. Clinical chemistry and laboratory medicine 58 (2020): 1116-1120.
- Tavazzi G, Pellegrini C, Maurelli M, et al. Myocardial localization of coronavirus in COVID-19 cardiogenic shock. European journal of heart failure 22 (2020): 911-915.
- Clerkin KJ, Fried JA, Raikhelkar J, et al. COVID-19 and Cardiovascular Disease. Circulation 141 (2020): 1648-1655.
- Tam CF, Cheung KS, Lam S, et al. Impact of Coronavirus Disease 2019 (COVID-19) Outbreak on ST-Segment-Elevation Myocardial Infarction Care in Hong Kong, China. Circulation Cardiovascular quality and outcomes 13 (2020): e006631.
- Stefanini GG, Azzolini E, Condorelli G. Critical Organizational Issues for Cardiologists in the COVID-19 Outbreak: A Frontline Experience From Milan, Italy. Circulation 141 (2020): 1597-1599.
- Garcia S, Albaghdadi MS, Meraj PM, et al. Reduction in ST-Segment Elevation Cardiac Catheterization Laboratory Activations in the United States During COVID-19 Pandemic. Journal of the American College of Cardiology 75 (2020): 2871-2872.
- Rodríguez-Leor O, Cid-Álvarez B, Ojeda S, et al. Impacto de la pandemia de COVID-19 sobre la actividad asistencial en cardiología intervencionista en España. REC Interv Cardiol 2 (2020): 82-89.
- Trabattoni D, Montorsi P, Merlino L. Late STEMI and NSTEMI patients’ emergency calling in CoVID-19 outbreak. Canadian Journal of Cardiology 36 (2020): 1161. e7- 1161. e8.
- Mountantonakis SE, Saleh M, Coleman K, et al. Out-of-Hospital Cardiac Arrest and Acute Coronary Syndrome Hospitalizations During the COVID-19 Surge. Journal of the American College of Cardiology 76 (2020): 1271-1273.
- Baldi E, Sechi GM, Mare C, et al. Out-of-hospital cardiac arrest during the Covid-19 outbreak in Italy. New England Journal of Medicine 383 (2020): 496-498.
- Cardiology JoI. SCAI and CAIC Announce the Formation of the North American COVID-19 ST-Segment Elevation Myocardial Infarction Registry (NACMI) (2020).
- Mahmud E, Dauerman HL, Welt FGP, et al. Management of Acute Myocardial Infarction During the COVID-19 Pandemic: A Position Statement From the Society for Cardiovascular Angiography and Interventions (SCAI), the American College of Cardiology (ACC), and the American College of Emergency Physicians (ACEP). Journal of the American College of Cardiology 76 (2020): 1375-1384.
- Chu CM, Cheng VC, Hung IF, et al. Role of lopinavir/ritonavir in the treatment of SARS: initial virological and clinical findings. Thorax 59 (2004): 252-256.
- Little P. Non-steroidal anti-inflammatory drugs and covid-19. BMJ (Clinical research ed) 368 (2020): m1185.
- Indications for ACE inhibitors in the early treatment of acute myocardial infarction: systematic overview of individual data from 100,000 patients in randomized trials. ACE Inhibitor Myocardial Infarction Collaborative Group. Circulation 97 (1998): 2202-2212.
- Chieffo A, Stefanini GG, Price S, et al. EAPCI position statement on invasive management of acute coronary syndromes during the COVID-19 pandemic. European heart journal 41 (2020): 1839-1851.
- Stefanini GG, Montorfano M, Trabattoni D, et al. ST-Elevation Myocardial Infarction in Patients With COVID-19: Clinical and Angiographic Outcomes. Circulation 141 (2020): 2113-2116.
- Daniels MJ, Cohen MG, Bavry AA, et al. Reperfusion of ST-Segment-Elevation Myocardial Infarction in the COVID-19 Era: Business as Usual?. Circulation 141 (2020): 1948-1950.