Alexei Konoplev*, Yoshifumi Wakiyama, Yasunori Igarashi, Kenji Nanba
Institute of Environmental Radioactivity, Fukushima University, Kanayagawa 1, Fukushima, Japan
*Corresponding Author: Alexei Konoplev, Institute of Environmental Radioactivity, Fukushima University, Kanayagawa 1, Fukushima, Japan
Received: 20 October 2023; Accepted: 30 October 2023; Published: 07 November 2023
Fukushima Dai-ichi Nuclear Power Plant (FDNPP) accident in March 2011 led to extensive environmental contamination by radiocesium isotopes 134Cs (half-life T1/2=2.06 years) and 137Cs (T1/2=30.17 years). Numerous research studies of environmental behavior of radiocesium in the context of geoclimatic conditions of Japan were undertaken. A lot of thought was given to what makes Fukushima environmental impacts different from previous nuclear disasters. This review paper summarizes key findings of post-Fukushima studies of radiocesium fate and transport in abiotic soil-water environment and discusses some challenges for future research. After the Fukushima accident scientific evidence was obtained to confirm that radiocesium behavior in the environment is governed by its speciation in fallout and site-specific environmental characteristics. Given strong binding of Fukushima-derived 137Cs to soil and sediment particles, its potential bioavailability appeared to be reduced. Incorporation of the deposited 137Cs in glassy hot microparticles insoluble in water and slowly decomposing in the environment was another salient feature. The Fukushima contaminated areas are noted by relatively high annual precipitation and steep slopes, resulting in significant erosion and intensive radiocesium wash-off, especially during devastating typhoons. Extreme floods during typhoons Etou in 2015 and Hagibis in 2019 caused major redistribution of 137Cs on river watersheds and floodplains, and, in some cases, natural self-decontamination. Significantly, for all special features and characteristics of Fukushima areas, the recently obtained knowledge is in line with the basic perceptions of modern environmental chemistry regarding radionuclide behavior in the environment. Challenges for future research include, to name a few, tackling radiocesium leaching from glassy hot particles and studying dynamics of radiocesium in the environment over long term, better understanding of dissolved radiocesium seasonality in water bodies and addressing radiocesium remobilization from rivertransported sediments at the freshwater-seawater interface.
Fukushima; Radiocesium; Speciation; Fate; Transport; Selfpurification; Challenges
Fukushima articles; Radiocesium articles; Speciation articles; Fate articles; Transport articles; Self-purification articles; Challenges articles
1. Introduction
The Great East Japan Earthquake and the following tsunami on 11 March 2011 caused the accident at FDNPP which led to extensive local soil contamination by 134Cs (half-life T1/2=2.06 years) and 137Cs (T1/2=30.17 years). Radiocesium (r-Cs) deposition north-west of the NPP resulted in a trace of contamination 50-70 km long and 20 km wide [1-4]. The initial ratio of 134Cs/137Cs isotopes in the Fukushima fallout was about one [2,5]. The contribution of 134Cs to the radioactive contamination and local air dose rate, as compared to 137Cs, decreased over time due to its more rapid decay [6,7].
The behavior of accidentally released r-Cs in the environment is determined by speciation in the fallout and site-specific environmental characteristics which are key for its mobility and bioavailability [8-10]. The areas of Japan contaminated as a result of FDNPP accident are characterized by wet monsoon climate with highly variable total annual precipitation (1200-1800 mm/yr). Less than 5-7% of precipitation falls as snow. Extreme rains with precipitation amount > 200 mm event occur once every 5-7 years according to meteorological observations. Several rains >50 mm fall each year, most frequently during the monsoon season between June and October. Some heavy rains can however occur in April-May, when soil surface on cultivated fields is less protected by vegetation [11]. Occurrence of high flow events in rivers especially during typhoon’s season facilitates r-Cs wash-off from contaminated catchments and its lateral migration [7,12-16].
The geology of the contaminated territory is highly heterogeneous, and the mountains serve as a partition between the Abukuma river valley, the largest in the region, and the ocean. The proportion of clays is 20-30%. These mountains are 1200-1300 m high and folded metamorphic and sedimentary rocks with numerous magmatic intrusions. Parent rock materials in the Fukushima contaminated areas are primarily granites and volcanic ashes that are subject to physico-chemical weathering in the humid monsoon climate conditions. In the region there are several active and dormant volcanoes; hot springs of different geochemical composition are abundant. Soil diversity is great due to the combination of mountain rocks of different lithological composition, intense weathering and denudation from high seismicity, and the steep inclination of mountain slopes. The interfluve areas include brown soils (under beech forest), ashy-volcanic, rich in humus, acidic allophonic (andosol) and leached brown soils. The valleys floors are used mainly for growing rice and are represented by alluvial soils strongly modified as a result of many years land use. Undisturbed alluvial soils occur on the leveed parts of river valleys and along the canalized parts of stream channels typical of intermountain depressions. The arable lands, mainly paddy fields, occupy about 12% of the total territory in the region, and occur primarily on extensive depressions and piedmont plain [17,18]. After the Fukushima accident, cultivation on arable land with high radiocesium deposition levels has been banned and decontamination activities have been implemented to remove contaminated soil layers [19,20].
The contaminated territory of Fukushima Prefecture is characterized by an expansive and differentiated hydrographic network, dominated by the largest river of the area – Abukuma. Other rivers include Ukedo, Niida, Uda, Mano, Ota, Odaka, Maeda, Kuma, Tomioka, Ide, Kido, Natsui and Same with their tributaries [21,22]. All these rivers ultimately end in the Pacific Ocean. Thus, the river catchments contaminated from the FDNPP accident became a long-term source of secondary contamination of water bodies by surface runoff and radiocesium flux to the Ocean [23,24]. Moreover, surface runoff and river transport results in the transfer of radiocesium from contaminated evacuated areas to cleaner populated regions, and the settling of radiocesium in bottom sediments of river dam reservoirs and on floodplains (mostly during rainy seasons) [7,25].
The objective of this review paper is to synthesize key findings regarding behavior of Fukushima-derived r-Cs in soil-water environment including catchment soils, sediments, rivers, ponds and dam reservoirs as well as to formulate relevant challenges for post-Fukushima environmental radioactivity studies in the future.
2. Radiocesium Speciation in Deposition and its Transformation with Time
2.1 Release of hot microparticles following FDNP accident
Immediately after the accident it was hypothesized that accidentally released r-Cs was transported in the atmosphere and deposited as part of sulfate aerosol particles of 0.5-0.6 μm diameter. R-Cs in these particles was assumed water-soluble and washable by precipitation [26]. Yet, water-insoluble spherical glassy hot particles of a few micrometers in diameter were discovered later as far as 170 km from the FDNPP, containing, apart from r-Cs, uranium, and other elements representative of reactor materials [27,28]. Later, Igarashi et al. [29] reported about determination of Pu isotopes in hot particles derived from Unit 1 of the FDNP. Similar particles have been identified by Niimura et al. [30] using autoradiography of soils, plants, and mushrooms. Near the FDNPP, even coarser particles (up to hundreds of µm) were identified with higher r-Cs activity (sometimes more than 1000 Bq/particle) and irregular shape [29]. These radiocesium-bearing microparticles (CsMPs) are primarily composed of silicates [31], and most likely were the product of molten core-concrete interaction during the accident, which was confirmed by laboratory experiments [32].
In terms of r-Cs fate and transport, it is important that CsMPs are insoluble in water and persistent in the environment [33,34]. For adequate modeling and prediction of Fukushima-derived r-Cs behavior in the environment, it is necessary to know the fraction of CsMPs activity in the total radiocesium release and deposition at different locations and the rate of radiocesium leaching from CsMPs due to decomposition [10].
Ikehara et al. [35,36] succeeded to characterize quantitatively the content of CsMPs in soils at different directions from the FDNPP. They found that CsMPs account for a significant fraction of FDNPP-deposited r-Cs. The number of CsMPs in surface soils at various directions, distances and deposition levels varied from few to more than 300 particles/g, and radioactivity fraction of CsMPs from the total deposition was in range 15-80%. Occurrence of CsMPs in soils and sediments can substantially impact the r-Cs solid-liquid distribution in the soil-water environment [37]. However, the variability of CsMPs fraction in deposition in a relatively wide range complicates accounting for CsMPs in assessment of mobility and bioavailability of r-Cs in the soil-water environment and its dynamics.
Miura et al. [33] discovered CsMPs in suspended sediments collected from Kuchibuto River (right bank tributary of the Abukuma River flowing from the most contaminated section of the Abukuma River catchment) during 2011-2016. The fraction of radiocesium incorporated in CsMPs to the total radiocesium in the sediments was found to be up to 67%.
2.2 Transformation of r-Cs chemical forms in the environment
After the deposition, the initial chemical forms of radionuclides are subject to transformation [10,38-40]. With CsMPs disintegrated in the environment due to weathering, r-Cs incorporated in these particles transfers to the solution [41,42]. In the solution, radiocesium is sorbed by the soil and sediment particles by ion exchange on selective frayed edge sites (FES) and regular exchange sites (RES) [43-45]. Exchangeably sorbed r-Cs is fixed by micaceous clay minerals, i.e., transferred into nonexchangeable r-Cs, due to the replacement of the interlayer K-cations by Cs-cations [46-50]. The mechanism of r-Cs fixation consists of the replacement of interlattice K+ by Cs+ ions due to the collapse of the expanded edges of the mineral’s crystal interlayers and/or the slow long-term solid-state diffusion of Cs+ ions through the interlayer inside the particle [38,51,52]. Data about long-term dynamics of r-Cs chemical forms in the soil and sediments are indicative of the existence of the remobilization process, which is the reverse of fixation [8,10,53-56].
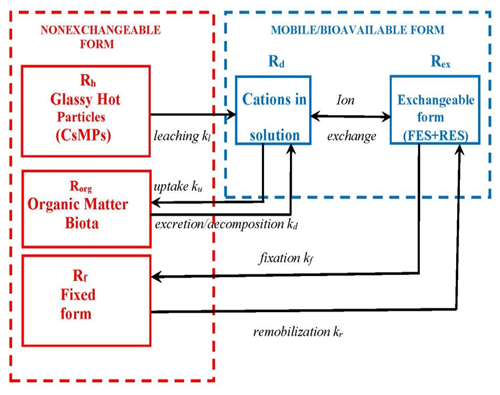
Radiocesium can be uptaken from solution by biota and reversely be released due to excretion or decomposition of dead organic matter [57-60]. Rates of these processes are variable and highly uncertain. However, in many cases they do not play critical role in r-Cs fate and transport [61]. Conceptual model of all major transformation processes for Fukushima-derived r-Cs chemical forms in the environment is presented in Figure 1.
2.3 Solid-liquid distribution of r-Cs in the soil-water environment
Solid-liquid distribution of r-Cs is a governing factor for its fate and transport in the soil-water environment. It is characterized by the apparent distribution coefficient Kd (L/kg) which is the ratio of the particulate radionuclide activity concentration [R]p (Bq/kg) to its dissolved activity concentration [R]d (Bq/L) at equilibrium [62]:

[R]p includes the r-Cs embedded in CsMPs (Rh), the exchangeably sorbed r-Cs (Rex), radiocesium fixed by clay minerals (Rf), and organically bound r-Cs (Rorg) (see Figure 1). The exchangeable r-Cs occurs at instantaneous ion-exchange equilibrium with the dissolved r-Cs, whereas the non-exchangeable form does not take part in the r-Cs exchange with the solution in immediate term. Therefore, it is worth using the exchangeable distribution coefficient , which is the ratio of the exchangeable radionuclide activity concentration in soil or sediments [R]ex to its activity concentration in solution at equilibrium [R]d [8,54]:
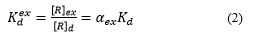
where is the exchangeable fraction of r-Cs in soil or sediments, and is the apparent (or total) distribution coefficient.
Soon after the FDNPP accident it was discovered that Fukushima-derived r-Cs is strongly bound to soil and sediment particles, and its apparent distribution coefficient Kd in Fukushima rivers is at least order of magnitude higher than in pre-Fukushima observations [22,37,63-65]. This fact was then confirmed by longer term studies [61,66-69]. Table 1 presents summary of typical available data on 137Cs distribution in suspended sediment-water system of rivers in Fukushima contaminated areas. The strong binding of r-Cs with soils and sediments seems to be associated with two factors: 1) relatively high fraction of micaceous clay minerals in Fukushima soils and sediments capable to selectively sorb and fix radiocesium [48,49,70-79] and 2) occurrence of high proportion of insoluble in water and persistent in the environment glassy hot particles CsMPs in FDNPP release [10,33]. It should be mentioned here that nuclear weapons testing derived r-Cs was not characterized by elevated values of Kd in rivers of Japan [80,81], which also confirms the role of FDNP-derived CsMPs for r-Cs solid-liquid distribution in Fukushima rivers.
|
River-site |
Observation period |
Mean value |
Reference |
|
Abukuma River-Kuroiwa |
2012-2020 |
(6.5±3.0) × 105 |
Igarashi et al. [63] |
|
Kuchibuto River upstream |
2011-2016 |
(4.9±2.1) × 105 |
Taniguchi et al. [68] |
|
Ukedo River downstream |
2015-2018 |
(2.2±0.3) × 105 |
Nakanishi and Sakuma [67] |
|
Ukedo River at Ogaki dam inflow |
2014-2019 |
(6.3±2.0) × 105 |
Funaki et al. [69] |
|
Kodeya River at Ogaki dam inflow |
2014-2019 |
(8.6±2.1) × 105 |
Funaki et al. [69] |
|
Ukedo River at Ogaki dam outflow |
2014-2019 |
(4.5±1.8) × 105 |
Funaki et al. [69] |
|
Takase River (Ukedo River system) |
2011-2016 |
(6.6±1.2) × 105 |
Taniguchi et al. [68] |
|
Ohta River downstream |
2015-2018 |
(2.4±0.6) × 105 |
Nakanishi and Sakuma [67] |
|
Niida River Haramachi |
2011-2016 |
(6.3±2.5) × 105 |
Taniguchi et al. [68] |
|
Hiso River (Niida River system) |
2011-2020 |
(4.6±3.0) × 105 |
Ueda et al. [70] |
|
Wariki River (Niida River System) |
2011-2020 |
(7.7±6.3) × 105 |
Ueda et al. [70] |
Table 1: 137Cs apparent distribution coefficient Kd (L/kg) in suspended sediment-water system of rivers in Fukushima contaminated areas.
Due to the fact that river water differs from marine water by hydrochemistry an interesting feature is observed such as Kd jump on the “freshwater-marine water” interface. The point is that freshwater differs markedly from seawater in terms of cation’s concentrations in solution and, in the first place, concentration of potassium, which is the key competitor of r-Cs for sorption sites FES. On the interface between fresh and coastal waters, the apparent Kd value of 137Cs jumps down by two orders of magnitude due to a sharp increase of potassium concentration from several mg/L in rivers to about 350-400 mg/L in coastal waters [82-84]. An obvious increase in potassium concentration also occurs from upstream to the mouth in a number of rivers in Fukushima contaminated areas. In Abukuma River, as an example, [K+] changed from 3.5 mg/L upstream to 44 mg/L at the mouth at tide, or in Maeda River [K+] changed from 0.7 mg/L upstream to 11.5 mg/L at the mouth in 2018 [84]. This leads to enhanced ion exchange desorption and remobilization of r-Cs from sediments to solution in the coastal areas of the Ocean. As a result, more than 95% of radiocesium in the ocean occurs in solution [85,86], while in rivers and other freshwater bodies a major part of radiocesium is bound to sediments [24,37,61,67,87]. A massive delivery of contaminated suspended material from accidental zone catchments by rivers during extreme floods in the course of typhoons causes an intense desorption and remobilization of 137Cs and its concentration increase in estuary [86].
2.4 Leaching of r-Cs due to CsMPs weathering
The processes of decomposition of CsMPs are difficult to model due to diversity of their size, shapes, and chemical characteristics [10,41]. Therefore, an integral parameter is often used such as the first order rate constant kl (yr-1) accounting for the rate of radionuclide leaching from hot particles, i.e. radionuclide transfer from hot particle to solution [10,88]. It can be expected that r-Cs leaching from CsMPs is very slow process. At the same time, when released from CsMPs, r-Cs in the soil-water environment is fairly quickly fixed by clay minerals of soils or sediments [47]. Based on laboratory experiments on r-Cs leaching from individual CsMPs by Okumura et al. [41] average values of rate constants of r-Cs leaching from CsMPs were estimated as kl ≥ 0.043 yr-1 in freshwater, and kl ≈ 0.44 yr-1 in seawater [10].
The rate constant can also be estimated from long-term monitoring data indirectly. Monitoring of 137Cs in heavily contaminated ponds in the vicinity of FDNPP revealed a gradual decline of its apparent solid-liquid distribution coefficient Kd from 2015 to 2019 [42], which is not typical of r-Cs dynamics in water bodies in mid- and long-term [23]. The initial and intermediate phases after the Chernobyl accident, and after nuclear weapon tests (NWT), were characterized by a slight increase of r-Cs apparent Kd in soils and sediments as a result of its fixation or aging [8,53,55]. Assuming that the Kd decline trend is associated with gradual remobilization of 137Cs due to leaching from CsMPs in the ponds kl was estimated to be in the range 0.12-0.18 yr-1 [42]. A similar trend for the radiocesium apparent Kd in 2012-2020 with the rate constant kl=0.05 yr-1 has been recently reported for suspended sediment-water system in Abukuma River at Fukushima city (Kuroiwa) [61]. Thus, the estimates based on long-term monitoring data are consistent with those derived from laboratory experiments of Okumura et al. [41] with individual CsMPs. It can be expected that kl obtained for natural freshwaters is higher than in laboratory experiments with pure water and lower than with marine water. This applies to the estimates presented above.
- Fixation of r-Cs by clay minerals and remobilization
As it appears from laboratory experiments and field observations with a variety of soils and sediments [47,54,55,89-91] the timescale of radiocesium fixation equals to weeks or months (kf = 4-20 yr-1), depending on environmental conditions, while the timescale of remobilization can be up to a few years (kr = 0.4-2 yr-1). Based on data on speciation of Fukushima-derived r-Cs [78,79,92,93], it is expected that these processes are characterized by similar rates in Fukushima soils and sediments [10].
3. Natural attenuation of radioactive contamination after FDNPP accident
3.1 Vertical and lateral redistribution of radiocesium in catchment soils
Processes of natural attenuation besides radioactive decay do not reduce the total amount of radionuclides in the environment, rather they affect radionuclide distribution over space and time [94]. Physical processes involved in natural attenuation (advection, diffusion, dispersion) dilute radionuclides in the environment or partially remove/relocate and spread them (wash-off, erosion, sedimentation and river transport) [7]. Processes of r-Cs vertical and lateral migration lead to gradual reduction in contamination of catchment soil, particularly its top layer [95-98]. This, in turn, results in a gradual decrease of r-Cs concentrations in both particulate and dissolved forms of surface runoff and river water [23,61,87].
Climate and geomorphological conditions essentially influence the rate of natural attenuation processes [7]. Fukushima’s watersheds are mostly hilly with steep slopes. Annual precipitation is relatively high with annual averages of about 1500 mm/year for Fukushima according to the Japan Meteorological Agency [11,65].
After deposition of radionuclides on the ground surface, over time the contamination migrates down through the soil profile [6,10,95,97,98]. The dynamic pattern of vertical distribution of radionuclides in soil is critical from the standpoint of external dose rate, availability of radionuclides for transfer to surface runoff and wind resuspension in the boundary atmospheric layer, availability of radionuclides for root uptake by plants and percolation to groundwater. Radionuclides migrate vertically in solution and as colloids with infiltration water flow or attached to fine soil particles. Transport of r-Cs in solution by infiltration is slower than the water flow because of its sorption-desorption and fixation on soil particles [99]. Fine soil particles containing radiocesium can move by penetrating through pores, cracks and cavities, as well as with infiltration flow (lessivage), and as a result of vital activity of plants and biota (bioturbation) [58,96]. Nevertheless, the vertical migration of radionuclides in undisturbed soils can be described by the advection-dispersion equation [57, 99,100]. Simultaneous solution of respective equations written separately for specific radiocesium chemical forms in soil Ri (Rh; Rorg; Rf ; Rex and Rd - see Figure 1) with allowance for their transformation [54,89,101] is the most accurate way of representing 137Cs migration by the advection-dispersion model:
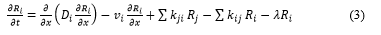
with the initial conditions
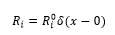
and boundary conditions
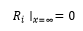
where Di and vi are the effective dispersion coefficient and effective advective velocity for each chemical form i, respectively; kij and kji are the rate constants of respective transformation process; and λ is the rate constant of radioactive decay equal 0.023 yr-1 for 137Cs.
Importantly, most vertical distributions of Fukushima-derived r-Cs demonstrated maximum of its activity concentrations in the topsoil layer for undisturbed land [6,95,97,98]. This means that 137Cs vertical distribution can be roughly approximated by simple diffusion equation with effective dispersion coefficient varying mostly in the range 1-9 cm2/yr [65,95,96].
Vertical distribution of r-Cs in floodplain soils differs significantly from that in undisturbed catchment soils because of erosion of topsoil and/or accumulation of sediments during floods [7,17,65]. Significant floods occur in Fukushima Prefecture during typhoon seasons, usually from mid-August to October. A very rare extreme floods occurred in September 2015 (Typhoon Etau) and in October 2019 (Typhoon Hagibis). The probability of such extreme floods in Fukushima contaminated area can be estimated at 4-5% per year [7]. During the floods, water flows washed out unsupported sections of stream banks. Also, some landslides occurred, mainly on very steep slopes. Sediments originated from the bank erosion, and landslides have very low concentration of 137Cs because of high proportion of material from deep soil layers. Therefore, extreme flood events in Fukushima contaminated areas very often cause substantial natural decontamination of river floodplains because of erosion of contaminated particles from the top layer and additional burying contaminated surface particles by deposited clean sediments [15,17]. In many cases this is followed by significant drop of air dose rate. Estimated rate constants of dose rate reduction for the sites without contaminated sediments accumulation in 2016 were in range of 0.2-0.4 year-1 [7]. Therefore, typhoons passing on Fukushima contaminated area result in relatively fast and efficient natural attenuation.
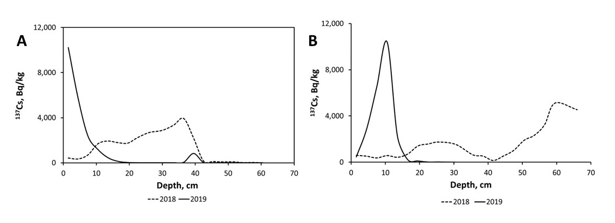
At the same time, in case of extreme floods on relatively large rivers, sediments with relatively high 137Cs concentrations can deposit on floodplain downstream influx of tributaries flowing through areas with elevated 137Cs levels [17]. This was the case after Typhoon “Hagibis” in October 2019 on the sections of Abukuma River floodplain downstream of Kuchibuto river catchment characterized by elevated depositions of 137Cs. Figure 2 shows vertical profiles of 137Cs for floodplain sediments of Abukuma River at two monitoring sites Fukushima (Kuroiwa) and Fushiguro located downstream of the mouth of the Kuchibuto River, before Typhoon “Hagibis” in 2018 and right after it in November 2019. The top layer of the floodplain sections was contaminated by sediments deposited after typhoon’s flood, while previous top layer sediments were removed by powerful water flow during the typhoon. It can be suggested that during the typhoon “Hagibis” the sediments from contaminated Kuchibuto River catchment were transported and deposited onto Abukuma River floodplain downstream [84].
Figure 2: Depth distribution of 137Cs in accumulation zones of floodplain sediments of Abukuma River at sites located downstream for the mouth of its right side tributary Kuchibuto River flowing through the FDNPP contaminated zone – Fukushima (at Kuroiwa) and Fushiguro, based on the data from Golosov et al. [17].
Accounting for soil erosion and sediment accumulation within river catchment and in particular, river floodplain, is key for predicting redistribution of radioactive contamination after the FDNPP accident on the contaminated territories, as well as for decision making about their remediation and clean-up. In many cases monitored natural attenuation along with appropriate restrictions can be the most optimal option for water remediation in Fukushima contaminated areas [7,94].
- Radiocesium accumulation in bottom sediments of dam reservoirs
Knowing temporal changes of 137Cs particulate concentrations in rivers is critically important for better understanding and prediction of radionuclide behavior in the soil-water environment. In the absence of systematic monitoring data, this gap can be filled by reconstructing particulate 137Cs concentrations based on 137Cs depth distribution in the bottom sediment column. Bottom sediments in dam reservoirs represent the final chain in the geological cycle of the matter within a river basin [102,103]. In the Fukushima contaminated areas, at least 10 major dams are in operation to support agricultural and surface water management [104,105]. Apparently, these reservoirs have trapped substantial fraction of transported radiocesium-contaminated sediments [106,107].
Depth distributions of radionuclides in bottom sediments of dam reservoirs can be used to reconstruct concentrations of strongly bound to sediment radionuclides occurring in rivers [108,109]. Sediments depositing in deep-water zones of reservoirs are delivered from the river and therefore they can serve to retrieve time changes of radionuclide concentrations in the river [25]. For reconstruction, however, it is required that mixing of sediments after deposition of radionuclides on the bottom be negligible, so that the radionuclide concentration in each individual sediment layer correspond to concentration in river suspended sediments during the respective time interval. Another requirement is that radionuclide should be strongly bound by sediments (Kd >> 104 L/kg) [25,109] which is valid for Fukushima-derived r-Cs [37].
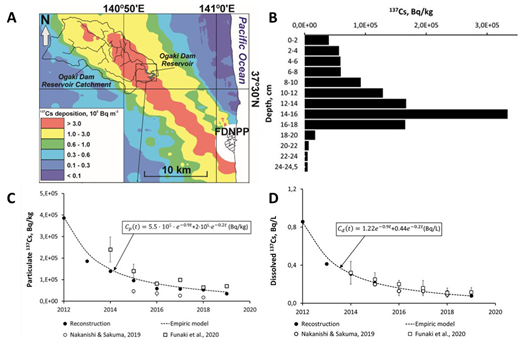
Figure 3. Reconstruction of time changes in 137Cs activity concentrations in the Ukedo River based on its depth distribution in Ogaki dam reservoir’s bottom sediments. A – location of Ukedo River and Ogaki dam in FDNPP contaminated area; B - 137Cs depth distribution profile for bottom sediment core from the Ogaki dam reservoir taken for reconstruction; C - time dependence of reconstructed particulate 137Cs activity concentration in the Ukedo river versus monitoring data [66,68] and approximation by the empirical two-exponential model; D - time dependence of reconstructed dissolved 137Cs activity concentration in Ukedo River versus monitoring data [66,68] and approximation by the empirical two-exponential model.
This approach was used for bottom sediments core from accumulation zone of Ogaki Dam at Ukedo River [25]. Figure 3 presents time dependencies of particulate and dissolved 137Cs in Ukedo River after the Fukushima accident reconstructed on the basis of bottom sediments core collected in 2019 and their comparison with monitoring data [66,68] and with empirical two-exponential model [25]. Time changes of particulate and dissolved 137Cs activity concentration in Ukedo River, reconstructed from the depth distribution in bottom sediments of deep-water accumulation zone in Ogaki Dam reservoir, are in good agreement with monitoring data [66,68] and are adequately approximated by the simple empirical two-exponential model. This means that the described method can be used in the future to reconstruct r-Cs dynamics in rivers of Fukushima contaminated areas.
4. Time Changes of 137Cs in Freshwaters and their Modelling
Initial radioactive contamination of water bodies after the nuclear FDNPP accident was relatively high, owing to direct fallout onto the rivers and lakes surfaces [18,22,63,64,110]. The contamination of water bodies was then decreasing sharply due to fast processes of sorption and fixation of radionuclides to sediments, as well as sedimentation of particles to the bottom [10,24,61,66,67,111-113]. Yet, extensive territories contaminated due to the FDNPP accident continue to serve as a long-term source of r-Cs to natural waters and aquatic ecosystems. Wash-off driven by surface runoff is the primary pathway for contamination of water bodies in mid- and long-term [11,18,21,24,87,114,115].
Prior the Fukushima accident, temporal changes in 137Cs activity concentrations in rivers were often described by the empirical fitting model using a series of exponential functions [116-118]:

where c(t) is the current 137Cs activity concentration in river (for particulate 137Cs in Bq/kg, and for dissolved in Bq/m3); λ is the 137Cs decay rate constant equal 0.023 yr-1; ki are empirically fitted rate constants; are fitting parameters, and t is time. This approach was followed by a number of researchers in post-Fukushima studies of r-Cs (both dissolved and particulate) dynamics in rivers [24,61,66-69].
Alternative way to model mid- and long-term dynamics of radiocesium in rivers is semiempirical diffusional approach [23,135]. The key assumption of the model is that the main source of suspended particles for surface runoff is the top layer of catchment soil, and r-Cs vertical distribution in the topsoil layer is described by the diffusional equation [96]. In this case time changes of 137Cs activity concentration in topsoil layer and thus in suspended sediments can be approximated by the following equation:
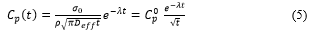
where σ0 is the initial average deposition of 137Cs on the catchment; Deff is the effective dispersion coefficient, averaged over the catchment area; is the radioactive decay rate constant; ρ is the average bulk density of the topsoil over the catchment; and t is time.
With allowance for equations (1) and (5), the time dependence of dissolved 137Cs concentration in a river can be approximated by the equation [23]:

The advantage of this approach is that the same equation can be used for middle- and long-term phases after a nuclear accident with the same values of physically based parameters which can be estimated or determined by field or laboratory studies. More simply, decay corrected particulate r-Cs activity concentrations in surface runoff and rivers are described by the inverse square root of time function.
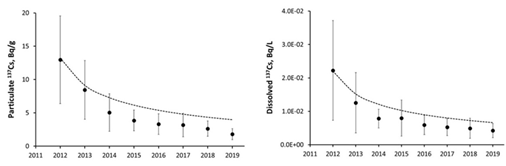
Figure 4 presents time changes of annual mean activity concentrations of dissolved 137Cs in Abukuma River at Fukushima city (Kuroiwa) in 2012-2019 [61] and their simulation by semi-empirical diffusional model. It should be noted that the 137Cs activity concentrations in rivers and lakes in Fukushima contaminated areas quite often decline a bit faster than predicted by the semi-empirical diffusional model [10]. There are two major potential reasons for the observed discrepancy and difference with modelling results. First, extensive remediation program implemented by Japanese government on the contaminated catchments after the accident has effectively reduced average 137Cs deposition on the catchment soils and especially decreased 137Cs content in topsoil layer [19,20,24]. Second, the basic processes of surface runoff development in Fukushima are quite specific due to relatively high intensity of precipitation, especially during typhoons, and high energy of surface runoff flows [11,105]. As a result, deeper soil layers become to be involved in exchange with surface runoff which causes faster decline of particulate and dissolved radionuclide concentration in surface runoff [7].
To characterize catchment-to-river transfer of radionuclides by surface runoff the particulate and dissolved wash-off ratios np and nd are widely used [18,22,23,61,87,115]:

where and are the particulate and dissolved radionuclide annual mean activity concentrations in river or surface runoff, respectively; σ is the current average radionuclide deposition on the catchment.
The wash-off ratios enable predicting the radionuclide wash-off from contaminated catchment and its concentration in rivers and other water bodies. To estimate the fraction of the radionuclide washed off in solution, the dissolved wash-off ratio is multiplied by the expected runoff depth for a given runoff event or period of interest. The fraction of radionuclide washed off with sediments is estimated by multiplying the particulate wash-off ratio by the predicted sediment yield during the runoff event or period of interest [119,120].
Time dependence of 137Cs particulate and dissolved wash-off ratios for mid- and long-term phases after the FDNP accident according to the semi-empirical diffusional model can be described as follows [121]:
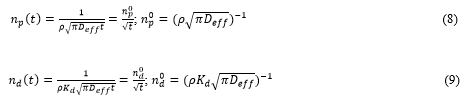
Decline of particulate wash-off ratio with time for Fukushima rivers can be described by diffusional model where =3.5-5.0 cm2/yr, whereas dissolved wash-off ratios follow the model at Kd=2.5×105 L/kg for Ukedo and Ohta rivers and at Kd=6×105 L/kg for Abukuma River at Fukushima, which values correspond to experimental data for these rivers [61,65,66].
5. Seasonal Variations of 137Cs in Contaminated Water Bodies
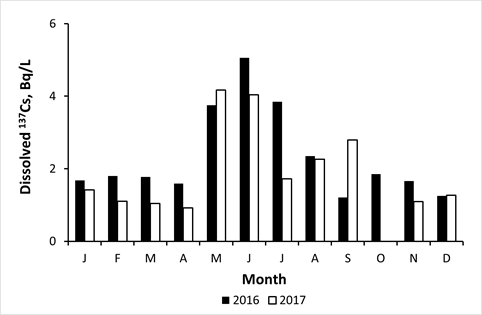
Rivers of the Fukushima contaminated areas demonstrated clear seasonal variations of dissolved 137Cs, showing pronounced minimum in winter and maximum in summer [61,66,87,122]. Similar seasonal variations were observed in Fukushima ponds, lakes, and dam reservoirs [42,123-125]. The amplitude of such variations, i.e. ratio of maximal summer and minimal winter dissolved 137Cs concentrations within the same year, is usually about 2-3 times. Figure 5 presents seasonal variations of dissolved 137Cs concentration in water of the pond Suzuuchi in Okuma town in 2016 and 2017 [42].
Conceivably, the seasonal changes in water temperature could have an impact on ion-exchange desorption of 137Cs which proceeds according to the Gibbs–Helmholtz equations for 137Cs exchangeable distribution coefficient [61,126]:

where ΔH is the enthalpy change for 137Cs desorption from sediments to solution at temperature T, and R is the gas constant. ΔH = - EA, where EA is Arrhenius activation energy [127-129]. The obtained estimates of EA were around 20 kJ/mol for Fukushima rivers [61,87] and ponds [42], which is consistent with results of laboratory experiments on sediments from Hanford site [126].
Seasonal variations of dissolved Chernobyl-derived 137Cs in lakes and ponds [113,130,131] were attributed to the variations in ammonium concentration in the water. Generally shallow and fast-flowing Fukushima rivers are characterized by negligible ammonium concentrations. At the same time, in the Fukushima stagnated water bodies such as lakes, ponds, and dam reservoirs the decomposition of organic matter in the reducing conditions of bottom-sediments could also have led to ammonium generation in pore water, which could have contributed to increased dissolved 137Cs in the water column in summer [42,123,124]. According to the r-Cs ion exchange theory [38,43,132,133], exchangeable distribution coefficient of 137Cs at constant water temperature depends on ammonium concentration as follows:

where RIPex(K) is exchangeable r-Cs interception potential, intrinsic property of soils and sediments accounting their ability to selectively and reversibly sorb r-Cs on frayed edge sites (FES) of micaceous clay minerals [113,132,133]; is selectivity coefficient of ammonium sorption on FES in relation to potassium. According to Wauters et al. [132,133], for more than hundred studied soils and sediments =5±2. Equation (11) clearly shows that looking for correlations of dissolved 137Cs activity concentration with single concentrations of potassium or ammonium in water is hopeless, when both these r-Cs competitors for sorption sites FES have comparable contribution in r-Cs desorption from sediments.
Combining equations (10) and (11) seasonal variations of and dissolved 137Cs activity concentration [137Cs]d in water body for mid- and long-term phases after the accident can be approximated by the following equations:
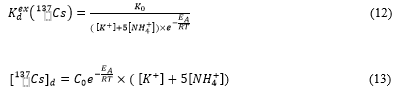
where K0 and C0 can be taken as a constant within the single year
Most likely, seasonality of dissolved 137Cs in Fukushima rivers is determined exclusively by changes of water temperature, since ammonium concentration in river water is normally negligible, and potassium concentration does not exhibit any regular seasonal variations. For ponds, lakes, and dam reservoirs the situation is more complicated when contribution of ammonium to 137Cs desorption is higher or comparable with contribution of potassium. It is expected that using of equations (12) and (13) should reduce the scattering of experimental points at the - and - plots for field observations especially in ponds and dam reservoirs [42,68,123,124]. However, applicability of equations (12) N (13) should be further investigated and tested in field observations.
6. Conclusions
The FDNPP accident in March 2011 led to extensive environmental contamination by radiocesium. Numerous studies of the radionuclide fate and transport in the environment in the context of geoclimatic conditions of Japan were carried out. The scientific evidence from conducted research confirmed that behavior of accidentally released radiocesium in the environment is governed by its speciation in fallout and site-specific environmental characteristics, even though the Fukushima-derived radiocesium has certainly distinct features and characteristics.
The Fukushima-derived 137Cs was found to be strongly bound to soil and sediment particles, which reduces its potential bioavailability. Also, in surface waters faster scavenging of radiocesium is observed and its transport primarily occurs on suspended material, which is important to bear in mind in modeling and prediction.
Up to 80% of the deposited 137Cs on the soil of the contaminated area were reported to be incorporated in glassy hot microparticles insoluble in water. These particles decompose in the environment very slowly, and long-term radiocesium leaching from these particles remains to be understood in future.
The frequently used in modeling parameter of apparent distribution coefficient Kd(137Cs) in the suspended sediments–water system of Fukushima rivers and ponds was shown to decrease with time after the accident. From the decrease in Kd estimates of the timescale of 137Cs leaching from glassy microparticles were obtained, ranging from 6 to 20 years, which is consistent with laboratory findings.
The high annual precipitation and steep slopes in Fukushima contaminated areas are conducive to significant erosion and intensive radiocesium wash-off, especially during devastating tropical storms. Typhoons Etou in 2015 and Hagibis in 2019 were reported to bring about major redistribution of 137Cs on river watersheds and floodplains, and in many cases natural self-decontamination was noticed.
With Fukushima dam reservoirs acting as traps, preventing radiocesium transport downstream, the 137Cs vertical distributions in bottom sediments of dam reservoir deep zones can be regarded as representing time changes in river water contamination.
he observed regular seasonal variations of dissolved 137Cs in water bodies (increase in summer and decrease in winter) can be attributed to two major factors: 1) temperature dependence of radiocesium desorption rate from sediments, and 2) generation of ammonium as a result of organic matter decomposition in anoxic conditions. Hence, a key mechanism of radiocesium seasonality in aquatic ecosystems is an important issue to be tackled in future.
Another challenge for contemporary environmental chemistry understands mechanisms and prediction of radiocesium remobilization from river-transported sediments at the freshwater-seawater interface.
Environmental radioactivity research following the Fukushima accident has made a major contribution to gaining greater knowledge about the processes of interest [134-136]. Nevertheless, unforeseen results can occur and require further investigation in future. Thus, new challenges are emerging, and environmental radiochemistry has yet to respond to them.
Acknowledgement
This research was supported by Environmental Radioactivity Research Center (ERAN) Projects I-23-11 and I-23-12.
References
- Chino M, Nakayama H, Nagai H, et al. Preliminary estimation of release amounts of 131I and 137Cs accidentally discharged from the Fukushima Daiichi nuclear power plant into the atmosphere. J Nucl Sci Technol 48 (2011): 1129-1134.
- Hirose K. 2011 Fukushima Dai-ichi nuclear power plant accident: summary of regional radioactive deposition monitoring results. J Environ Radioact 111 (2012): 13-17.
- MEXT (Ministry of Education, Culture, Sports, Science and Technology of Japan). Results of the (i) Fifth Airborne Monitoring Survey and (ii) Airborne Monitoring Survey Outside 80 km from the Fukushima Dai-ichi NPP (2012).
- Saito K, Tanihata I, Fujiwara M, et al. Detailed deposition density maps constructed by large-scale soil sampling for gamma-ray emitting radioactive nuclides from the Fukushima Dai-ichi Nuclear Power Plant accident. J Environ Radioact 139 (2015): 308-319.
- Chaisan K, Smith JT, Bossew P, et al. Worldwide isotope ratios of the Fukushima release and early-phase external dose reconstruction. Sci Rep 3 (2013): 2520.
- Malins A, Kurikami H, Nakama S, et al. Evaluation of ambient dose equivalent rates influenced by vertical and horizontal distribution of radioactive cesium in soil in Fukushima Prefecture. J Environ Radioact 151 (2016): 38-49.
- Konoplev A, Golosov V, Wakiyama Y, et al. Natural attenuation of Fukushima-derived radiocesium in soils due to its vertical and lateral migration. J Environ Radioact 186 (2018): 23-33.
- Beresford N, Fesenko S, Konoplev A, et al. Thirty years after the Chernobyl accident: what lessons have we learnt? J Environ Radioact 157 (2016): 77-89.
- Hirose K, Povinec PP. Ten years of investigations of Fukushima radionuclides in the environment: A review on process studies in environmental compartments. J Environ Radioact (2022): 251-252.
- Konoplev A. Fukushima and Chernobyl: similarities and differences of radiocesium behavior in the soil–water environment. Toxics 10 (2022): 578.
- Laceby JP, Chartin C, Evrard O, et al. Rainfall erosivity in catchments contaminated with fallout from the Fukushima Daiichi nuclear power plant accident. Hydrol Earth Syst Sci 20 (2016): 2467-2482.
- Evrard O, Chartin C, Onda Y, et al. Renewed soil erosion and remobilization of radioactive sediment in Fukushima coastal rivers after the 2013 typhoons. Sci Rep 4 (2014): 4574.
- Nagao S, Kanamori M, Ochiai S, et al. et al. Export of 134Cs and 137Cs in the Fukushima River systems at heavy rains by Typhoon Roke in September 2011. Biogeosciences 10 (2013): 6215-6223.
- Niida T, Wakiyama Y, Takata H, et al. A comparative study of riverine 137Cs dynamics during high-flow events at three contaminated river catchments in Fukushima. Sci Total Environ 821 (2022): 153408.
- Wakiyama Y, Konoplev A, Thoa N, et al. Temporal Variations in Particulate and Dissolved 137Cs Activity Concentrations in the Abukuma River During Two High-Flow Events in 2018. In: Nanba K, Konoplev A, Wada T (eds) Behavior of Radionuclides in the Environment III: Fukushima. SPRINGER Nature Singapore (2022): 153-176.
- Yamashiki Y, Onda Y, Smith HG, et al. Initial flux of sediment-associated radiocesium to the ocean from largest river impacted by Fukushima Daiichi Nuclear Power Plant. Sci Rep 4 (2014): 3714.
- Golosov V, Konoplev A, Wakiyama Y, et al. Erosion and redeposition of sediments and sediment-associated radiocesium on river floodplains (the Niida River basin and the Abukuma River as an example). In: Nanba K, Konoplev A, Wada T (eds) Behavior of Radionuclides in the Environment III: Fukushima. SPRINGER Nature Singapore (2022): 97-135.
- Konoplev A. Comparative analysis of radiocesium wash-off from contaminated watersheds as a result of the accidents at Fukushima Dai-ichi and Chernobyl NPPs. Geochemistry International 54 (2016): 522-528.
- Hardie SML, McKinley IG. Fukushima remediation: status and overview of future plans. J Environ Radioact 133 (2014): 75-85.
- Feng B, Onda Y, Wakiyama Y, et al. Persistent impact of Fukushima decontamination on soil erosion and suspended sediment. Nature Sustainability 5 (2022): 879-889.
- Evrard O, Laceby JP, Lepage H, et al. Radiocesium transfer from hillslopes to the Pacific Ocean after the Fukushima Nuclear Power Plant accident: A review. J Environ Radioact 148 (2015): 92-110.
- Yoshimura K, Onda Y, Sakaguchi A, et al. An extensive study of the concentrations of particulate/dissolved radiocaesium derived from the Fukushima Dai-ichi Nuclear Power Plant accident in various river systems and their relationship with catchment inventory. J Environ Radioact 139 (2015): 370-378.
- Konoplev A, Kanivets V, Zhukova O, et al. Mid- to long-term radiocesium wash-off from contaminated catchments at Chernobyl and Fukushima. Water Research 188 (2021): 116514.
- Onda Y, Taniguchi K, Yoshimura K, et al. Radionuclides from the Fukushima Daiichi Nuclear Power Plant in terrestrial systems. Nature Reviews Earth and Environ 1 (2020): 644-660.
- Konoplev A, Wakiyama Y, Wada T, et al. Reconstruction of time changes in radiocesium concentrations in the river of the Fukushima Dai-ichi NPP contaminated area based on its depth distribution in dam reservoir's bottom sediments. Environmental Research 206 (2022): 112307.
- Kaneyasu N, Ohashi H, Suzuki F, et al. Sulfate Aerosol as a Potential Transport Medium of Radiocesium from the Fukushima Nuclear Accident. Environ Sci Technol 46 (2012): 5720-5726.
- Adachi K, Kajino M, Zaizen Y, et al. Emission of spherical cesium-bearing particles from an early stage of the Fukushima nuclear accident. Sci Rep 3 (2013): 2554.
- Abe Y, Iizawa Y, Terada Y, et al. Detection of uranium and chemical state analysis of individual radioactive microparticles emitted from the Fukushima nuclear accident using multiple synchrotron radiation X-ray analyses. Anal Chem 88 (2015): 8521-8525.
- Igarashi Y, Kogure T, Kurihara Y, et al. A review of Cs-bearing microparticles in the environment emitted by the Fukushima Dai-ichi Nuclear Power Plant accident. J Environ Radioact 205-206 (2019): 101-118.
- Niimura N, Kikuchi K, Tuyen ND, et al. Physical properties, structure and shape of radioactive Cs from the Fukushima Daiichi Nuclear Power Plant accident derived from soil, bamboo and shiitake mushroom measurements. J Environ Radioact 139 (2015): 234-239.
- Satou Y, Sueki K, Sasa K, et al. Analysis of two forms of radioactive particles emitted during the early stages of the Fukushima Dai-ichi Nuclear Power Station accident. Geochemical J 52 (2018): 137-143.
- Laffolley H, Journeau C, Delacroix J, et al. Synthesis of Fukushima Daiichi Cs-bearing microparticles through molten core-concrete interaction in nitrogen atmosphere. Nuclear Materials and Energy 33: 101253.
- Miura H, Kurihara Y, Sakaguchi A, et al. Discovery of radiocesium-bearing microparticles in river water and their influence on the solid-water distribution coefficient (Kd) of radiocesium in the Kuchibuto River in Fukushima. Geochem J 52 (2018): 1-10.
- Miura H, Ishimaru T, Ito Y, et al. et al. First isolation and analysis of caesium-bearing microparticles from marine samples in the Pacific coastal area near Fukushima Prefecture. Sci Rep 11 (2021): 5664.
- Ikehara R, Suetake M, Komiya T, et al. Novel method of quantifying cesium-rich microparticles (CsMPs) in the environment from the Fukushima Daiichi nuclear power plant. Environ Sci Technol 52 2018: 6390-6398.
- Ikehara R, Morooka K, Suetake M, et al. Abundance and distribution of radioactive cesium-rich microparticles released from the Fukushima Daiichi Nuclear Power Plant into the environment. Chemosphere 241 (2020): 125019.
- Konoplev AV. Distribution of radiocesium of accidentally origin between suspended matter and solution in rivers: comparison of Fukushima and Chernobyl. Radiochemistry 57 (2015): 471-474.
- Konoplev A, Wakiyama Y, Wada T, et al. Behavior of Fukushima-Derived Radiocesium in the Soil–Water Environment: Review. In: Nanba K, Konoplev A, Wada T (eds) Behavior of Radionuclides in the Environment III: Fukushima. SPRINGER Nature Singapore (2022): 33-68.
- Salbu B, Lind OC, Skipperud L. Radionuclide speciation and its relevance in environmental impact assessments. J Environ Radioact 74 (2004): 233-242.
- Salbu B, Kashparov V, Lind OC, et al. Challenges associated with the behaviour of radioactive particles in the environment. J Environ Radioact 186 (2018): 101-115.
- Okumura T, Yamaguchi N, Dohi T, et al. Dissolution behavior of radiocesium-bearing microparticles released from the Fukushima nuclear plant. Sci Rep 9 (2019): 3520.
- Konoplev A, Wakiyama Y, Wada T, et al. Radiocesium distribution and mid-term dynamics in the ponds of the Fukushima Dai-ichi nuclear power plant exclusion zone. Chemosphere 265 (2021b): 129058.
- Cremers A, Elsen A, De Preter P, et al. Quantitative analysis of radiocesium retention in soils. Nature 335 (1988): 247-249.
- Fuller AJ, Shaw S, Peacock CL, et al. Ionic strength and pH dependent multi-site sorption of Cs onto a micaceous aquifer sediment. Applied Geochemistry 40 (2014): 32-42.
- De Koning A, Konoplev A, Comans R. Measuring the specific caesium sorption capacity of soils sediments and clay minerals. Applied Geochemistry 22 (2007): 219-229.
- Sawhney BL. Selective sorption and fixation of cations by clay minerals: a review. Clays and clay minerals 20 (1972): 93-100.
- Konoplev AV, Bulgakov AA, Popov VE, et al. Long-term investigation of 137Cs fixation by soils. Radiation Protection Dosimetry 64 (1996): 15-18.
- Nakao A, Thiry Y, Funakawa S, et al. Characterization of the frayed edge site of micaceous minerals in soil clays influenced by different pedogenetic conditions in Japan and northern Thailand. Soil Sci Plant Nutr 54 (2008): 479-489.
- Kogure T, Morimoto K, Tamura K, et al. XRD and HRTEM evidence for fixation of cesium ions in vermiculite clay. Chem Lett 41 (2012): 380-382.
- Fuller AJ, Shaw S, Ward MB, et al. Caesium incorporation and retention in illite interlayers. Applied Clay Science 108 (2015): 128-134.
- Comans RNJ, Haller M, De Preter P. Sorption of caesium on illite: nonequilibrium behaviour and reversibility. Geochim. Cosmochim Acta 55 (1991): 433-440.
- Comans RNJ, Hockley DE. Kinetics of caesium sorption on illite. Geochim. Cosmochim. Acta 56 (1992): 1157-1164.
- Pavlotskaya FI. Migration of radioactive products of global fallout in soils. Atomizdat, Moscow 270 (1974) (In Russian).
- Konoplev AV, Bulgakov AA, Popov VE, et al. Behaviour of long lived Chernobyl radionuclides in a soil water system. Analyst 117 (1992): 1041 1047.
- Smith JT, Comans RNJ. Modelling the diffusive transport and remobilisation of 137Cs in sediments: the effect of sorption kinetics and reversibility. Geokhim Cosmochim Acta 60 (1996): 995-1004.
- Konoplev AV, Bulgakov AA. Transformation of the forms of 90Sr and 137Cs soil and bottom deposits. At Energy 88 (2000): 56-60.
- Bulgakov AA, Konoplev AV, Shkuratova IG. Distribution of 137Cs in the topmost soil layer within a 30-km zone around the Chernobyl nuclear power plant. Pochvovedenie 9 (2000): 1149-1152 (In Russian).
- Bulgakov AA, Konoplev AV. Modelling of 137Cs vertical soil transfer by a tree root system. Radiation Biology. Radioecology 42 (2002): 556-560 (In Russian).
- Konoplev AV, Avila R, Bulgakov AA, et al. Quantitative assessment of radiocaesium bioavailability in forest soils. Radiochim. Acta 88 (2000): 789-792.
- Van der Stricht E, Kirchman R (Ed.). Radioecology. Radioactivity & Ecosystems. IUR, Belgium, 603 (2001).
- Igarashi Y, Nanba K, Wada T, et al. Factors Controlling the Dissolved 137Cs Seasonal Fluctuations in the Abukuma River Under the Influence of the Fukushima Nuclear Power Plant Accident. J Geophys Res Biogeosciences 127 (2022): 1-16.
- Handbook of parameter values for the prediction of radionuclide transfer in terrestrial and freshwater environments. Technical reports series No 472 Vienna (2010): 194.
- Ueda S, Hasegawa H, Kakiuchi H, et al. Fluvial Discharges of Radiocesium from Watersheds Contaminated by Fukushima Dai-ichi Nuclear Plant Accident, Japan. J Environ Radioact 118 (2013): 96-104.
- Sakaguchi A, Tanaka K, Iwatani H, et al. Size distribution studies of 137Cs in river water in the Abukuma Riverine system following the Fukushima Dai-ichi Nuclear Power Plant accident. J Environ Radioact 139 (2015): 379-389.
- Konoplev A, Golosov V, Laptev G, et al. Behavior of accidentally released radiocesium in soil-water environment: looking at Fukushima from a Chernobyl perspective. J Environ Radioact 151 (2016a): 568-578.
- Nakanishi T, Sakuma K. Trend of 137Cs concentration in river water in the medium term and future following the Fukushima nuclear accident. Chemosphere 215 (2019): 272-279.
- Taniguchi K, Onda Y, Smith HG, et al. Transport and redistribution of radiocesium in Fukushima fallout through rivers. Environ. Sci Technol 53 (2019): 1233912347.
- Funaki H, Sakuma K, Nakanishi T, et al. Reservoir sediments as a long-term source of dissolved radiocesium in water system; a mass balance case study of an artificial reservoir in Fukushima, Japan. Sci Total Environ 743 (2020): 140668.
- Ueda S, Hasegawa H, Ohtsuka Y, et al. Ten-year radiocesium fluvial discharge patterns from watersheds contaminated by the Fukushima nuclear power plant accident. J Environ Radioact 240 (2021): 106759.
- Fan QH, Tanaka M, Tanaka K, et al. An EXAFS study on the effects of natural organic matter and expandability of clay minerals on cesium adsorption and mobility. Geochim Cosmochim Acta 135 (2014): 49-65.
- Mukai H, Hatta T, Kitazawa H, et al. Speciation of radioactive soil particles in the Fukushima contaminated area by IP autoradiography and microanalysis. Environ Sci Technol 48 (2014): 13053-13059.
- Nakao A, Takeda A, Ogasawara S, et al. Relationship between paddy soil radiocesium interception potentials and physicochemical properties in Fukushima, Japan. J Environ Qual 44 (2015): 780-788.
- Nakao A, Nakao A, Ryoji T, et al. Aeolian-dust-derived micaceous minerals control radiocesium retention in andosols in Japan. Soil Sci Soc Am J 79 (2015): 1590-1600.
- Nakao A, Tomita M, Wagai R, et al. Asian dust increases radiocesium retention ability of serpentine soils in Japan. J Environ Radioact 204 (2019): 86-94.
- Mukai H, Motai S, Yaita T, et al. Identification of the actual cesium-adsorbing materials in the contaminated Fukushima soil. Appl. Clay Sci 121-122 (2016): 188-193.
- Mukai H, Hirose A, Motai S, et al. Cesium adsorption/desorption behavior of clay minerals considering actual contamination conditions in Fukushima. Sci Rep 6 (2016): 21543.
- Yamaguchi N, Tsukada H, Kohyama K, et al. Radiocesium interception potential of agricultural soils in northeast Japan. Soil Sci Plant Nutr 63 (2017): 119-126.
- Yamaguchi N, Hikono A, Saito T. Effects of zeolite and vermiculite addition on exchangeable radiocesium in soil with accelerated ageing. J Environ Radioact 203 (2019): 18-24.
- Fujii K, Yamaguchi N, Imamura N, et al. Effects of radiocesium fixation potentials on 137Cs retention in volcanic soil profiles of Fukushima forests. J Environ Radioact 198 (2019): 126-134.
- Hirose K, Aoyama M, Sugimura Y. Plutonium and cesium isotopes in river water in Japan. J Radioanal. Nuclear Chem 141 (1990): 191-202.
- Matsunaga T, Amano H, Yanase N. Discharge of dissolved and particulate 137Cs in the Kuji River, Japan. Applied Geochem 6 (1991): 159-167.
- Kusakabe M, Takata H. Temporal trends of 137Cs concentration in seawaters and bottom sediments in coastal waters around Japan: implications for the Kd concept in the dynamic marine environment. J Radioanal. Nuclear Chem 323 (2020): 567-580.
- Takata H, Wada T, Aono T, et al. Factors controlling dissolved 137Cs activities in coastal waters on the eastern and western sides of Honshu, Japan. Sci Total Environ 806 (2022): 151216.
- Konoplev A. Radioecology after Fukushima: lessons learned and challenges for the future. Proceedings of the 9th Annual Symposium of the IER, Fukushima University, Fukushima (2023).
- Aoyama M, Hamajima Y, Inomata Y, et al. Radiocaesium derived from the TEPCO Fukushima accident in the North Pacific Ocean: Surface transport processes until 2017. J Environ Radioact 189 (2018): 93-102.
- Takata H, Wakiyama Y, Niida T, et al. Importance of desorption process from Abukuma River’s suspended particles in increasing dissolved 137Cs in coastal water during river-flood caused by typhoons. Chemosphere 281 (2021): 130751.
- Nanba K, Moritaka S, Igarashi S. Dynamics of radiocesium in urban river in Fukushima-city. In: Nanba K, Konoplev A, Wada T (eds) Behavior of Radionuclides in the Environment III: Fukushima. SPRINGER Nature Singapore (2022): 137-152.
- Konoplev AV, Bulgakov AA. Kinetics of the leaching of 90Sr from fuel particles in soil in the near zone of the Chernobyl nuclear power plant. At Energy 86 (1999): 136-141.
- Abril JM, Barros H. Modelling the kinetic reactive transport of pollutants at the sediment-water interface. Applications with atmospheric fallout radionuclides. J Environ Radioact 242 (2022): 106790.
- Barros H, Abril JM. Kinetic box models for the uptake of radionuclides and heavy metals by suspended particulate matter: equivalence between models and its implications. J Environ Radioact 99 (2008): 146-158.
- Benes P, Picat P, Chernik M, et al. Kinetics of radionuclide interaction with suspended solids in modeling the migration of radionuclides in rivers. I. parameters for two-step kinetics. J Radioanal Nucl Chem 159 (1992): 175-186.
- Takeda A, Tsukada H, Nakao A, et al. Time-dependent changes of phytoavailability of Cs added to allophanic Andosols in laboratory cultivations and extraction tests. J. Environ Radioact 122 (2013): 29-36.
- Manaka T, Imamura N, Kaneko S,et al. Six-year trends in exchangeable radiocesium in Fukushima forest soils. J Environ Radioact 203 (2019): 84-92.
- IAEA (International Atomic Energy Agency). Applicability of Monitored Natural Attenuation at Radioactively Contaminated Sites. Technical Reports Series No. 445. IAEA, Vienna (2006): 105.
- Mishra S, Sahoo S, Bossew P, et al. Vertical migration of radio-cesium derived from the Fukushima Dai-ichi Nuclear Power Plant accident in undisturbed soils of grassland and forest. J Geochem Exploration 169 (2016): 163-186.
- Konoplev AV, Golosov VN, Yoschenko VI, et al. Vertical distribution of radiocesium in soils of the area affected by the Fukushima Dai-ichi nuclear power plant accident. Eurasian Soil Science 49 (2016): 570-580.
- Takahashi J, Tamura K, Suda T, et al. Vertical distribution and temporal changes of 137Cs in soil profiles under various land uses after the Fukushima Dai-ichi Nuclear Power Plant accident. J Environ Radioact 139 (2015): 351-361.
- Takahashi J, Onda Y, Hihara D, et al. Six-year monitoring of the vertical distribution of radiocesium in three forest soils after the Fukushima Dai-ichi Nuclear Power Plant accident. J Environ Radioact 210 (2019): 105811.
- Van Genuchten MT, Wierenga PJ. Solute Dispersion Coefficients and Retardation Factors. Methods of Soil Analysis. Part 1 Physical and Mineralogical Methods, Madison, Wisconsin USA (1986): 1025-1054.
- Smith JT, Hilton J, Comans RNJ. Application of two simple models to the transport of 137Cs in an upland organic catchment. Sci. Total Environ 168 (1995): 57-61.
- Konoplev AV, Golubenkov AV. Modeling of the vertical radionuclide migration in soil (as a result of a nuclear accident). Meteorol Gidrol 10 (1991): 62-68 (In Russian).
- Appleby PG. Tracking environmental change using lake sediments. In: Last WM, Smol JP (eds). Chronostratigraphic techniques in recent sediments. Developments in Paleoenvironmental Research Springer Netherlands (2002): 171-203.
- Funaki H, Yoshimura K, Sakuma K, et al. Evaluation of particulate 137Cs discharge from a mountainous forested catchment using reservoir sediments and sinking particles. J Environ Radioact 189 (2018): 48-56.
- Hayashi S, Tsuji H. Role and effect of a dam on migration of radioactive cesium in a river catchment after the Fukushima Daiichi Nuclear Power Plant accident. Global Environ Res 24 (2020): 105-113.
- Huon S, Hayashi S, Laceby JP, et al. Source dynamics of radiocesium-contaminated particulate matter deposited in an agricultural water reservoir after the Fukushima nuclear accident. Sci Total Environ 612 (2018): 1079-1090.
- Kurikami H, Kitamura A, Yokuda ST, et al. Sediment and 137Cs behaviors in the Ogaki Dam Reservoir during a heavy rainfall event. J Environ Radioact 137 (2014): 10-17.
- Yamada S, Kitamura A, Kurikami H, et al. Sediment and 137Cs transport and accumulation in the Ogaki dam of eastern Fukushima. Environ Res Lett 10 (2015): 1-9.
- Konoplev AV, Ivanov MM, Golosov VN, et al. Reconstruction of long-term dynamics of Chernobyl-derived 137Cs in the Upa River using bottom sediments in the Schekino reservoir and semi-empirical modelling. Proceedings of International Association of Hydrological Sciences 381 (2019): 95-99.
- Ivanov MM, Konoplev AV, Walling DE, et al. Using reservoir sediment deposits to reconstruct the longer-term fate of Chernobyl-derived 137Cs fallout in the fluvial system. Environmental Pollution 274 (2021): 116588.
- Nagao S, Kanamori M, Ochiai S, et al. Dispersion of Cs-134 and Cs-137 in river waters from Fukushima and Gunma prefectures at nine months after the Fukushima Daiichi NPP accident. Progr Nuclear Sci Technol 4 (2014): 9-13.
- Delmas M, Garcia-Sanchez L, Onda Y. Factors controlling the variability of 137Cs concentrations in 5 coastal rivers around Fukushima Dai-ichi power plant. J Environ Radioact 204 (2019): 1-11.
- Hayashi S, Tsuji H, Yumiko I. Effects of forest litter on dissolved 137Cs concentrations in a highly contaminated mountain river in Fukushima. Journal of Hydrology: Regional Studies 41 (2022): 101099.
- Konoplev AV, Kaminski S, Klemt E, et al. Comparative study of 137Cs partitioning between solid and liquid phases in Lakes Constance, Lugano and Vorsee. J. Environ. Radioact. 58 (2002): 1-11.
- Kitamura A, Yamaguchi M, Kurikami H, et al. Predicting sediment and cesium-137 discharge from catchments in eastern Fukushima. Anthropocene 5 (2014): 22-31.
- Wakiyama Y, Onda Y, Yoshimura K, et al. Land use types control solid wash-off rate and entrainment coefficient of Fukushima-derived 137Cs, and their time dependence. J Environ Radioact 210 (2019): 105990.
- Hilton J. Aquatic radioecology post Chernobyl – a review of the past and look to the future. In Freshwater and Estuarine Radioecology. In: Desmet G, Blust RJ, Comans RNJ, Fernandez JA, Hilton J, De Bettencourt A Eds., Elsevier: Amsterdam The Netherland (1997): 47-74.
- Monte L. A collective model for predicting the long-term behaviour of radionuclides in rivers. Sci Total Environ 201 (1997): 17-29.
- Smith JT, Belova NV, Bulgakov AA, et al. The “AQUASCOPE” Simplified model for predicting 89,90Sr, and 134,137Cs in surface waters after a large-scale radioactive fallout. Health Phys 89 (2005): 628-644.
- Borzilov VA, Konoplev AV, Revina SK, et al. An experimental study of the washout of radionuclides fallen on soil in consequence of the Chernobyl failure. Soviet Meteorol Hydrol 11 (1988): 27-33.
- Bulgakov AA, Konoplev AV, Popov VE, et al. Removal of long-lived radionuclides from soil by surface runoff near the Chernobyl nuclear power station. Soviet Soil Sci 23 (1991): 124-131.
- Konoplev AV, Kanivets VI, Zhukova OM, et al. Semi-empirical diffusional model of radionuclide wash-off from contaminated watersheds ant its testing using monitoring data for Fukushima and Chernobyl rivers. Geochemistry International 59 (2021c): 607-617.
- Tsuji H, Nishikiori T, Yasutaka T, et al. Behavior of dissolved radiocesium in river water in a forested watershed in Fukushima Prefecture. Journal of Geophysical Research: Biogeosciences 121 (2016): 2588-2599.
- Funaki H, Tsuji H, Nakanishi N, et al. Remobilisation of radiocaesium from bottom sediments to water column in reservoirs in Fukushima, Japan. Sci Total Environ 812 (2022): 152534.
- Matsuzaki S, Tanaka A, Kohzu A, et al. Seasonal dynamics of the activities of dissolved 137Cs and the 137Cs of fish in a shallow, hypereutrophic lake: Links to bottom-water oxygen concentrations. Sci Total Environ 761 (2021): 143257.
- Tsuji H, Funaki H, Watanabe M, et al. Effects of temperature and oxygen on 137Cs desorption from bottom sediment of a dam lake. Appl Goechem 140 (2022): 105303.
- Liu C, Zachara JM, Qafoku O, et al. Effect of temperature on Cs+ sorption and desorption in subsurface sediments at the Hanford Site, USA. Environ Sci Technol 37 (2003): 2640-2645.
- Semenov NN. Some problems of chemical kinetics and reactivity. Moscow, Academy of Science 686 (1958) (In Russian).
- Benson SW. The foundations of chemical kinetics (Advanced Chemistry). McGraw-Hill Book Company (1960).
- Laidler KJ. Chemical Kinetics 3rd Edition, Harper and Row (1987): 531.
- Ries T, Putyrskaya V, Klemt E. Long-term distribution and migration of 137Cs in a small lake ecosystem with organic rich catchment: A caser study of Lake Vorsee (Southern Germany). J Environ Radioact 198 (2019): 89-103.
- Kanivets V, Laptev G, Konoplev A, et al. Distribution and dynamics of radionuclides in the Chernobyl cooling pond. In: Konoplev A, Kato K, Kalmykov SN (Eds.) Behavior of Radionuclides in the Environment II: Chernobyl. SPRINGER Nature (2020): 349-405.
- Wauters J, Madruga MJ, Vidal M, et al. Solid phase speciation of radiocesium in bottom sediments. Sci. Total Environ 187 (1996): 121-130.
- Wauters J, Elsen A, Cremers, et al. Prediction of solid/liquid distribution coefficients of radiocesium in soils and sediments. Part one: A simplified procedure for the solid phase characterization. Appl Geochem 11 (1996): 589-594.
- Sakashita W, Miura S, Akama A, et al. Assessment of vertical radiocesium transfer in soil via roots. J Environ Radioact 222 (2020): 106369.
- Konoplev A, Kanivets V, Laptev G, et al. Long-term dynamics of the Chernobyl-derived radionuclides in rivers and lakes. In: Konoplev A, Kato K, Kalmykov SN (Eds.) Behavior of Radionuclides in the Environment II: Chernobyl. SPRINGER Nature (2020): 323-348.
- Nakao A, Ogasawara S, Sano O, et al. Radiocesium sorption in relation to clay mineralogy of paddy soils in Fukushima, Japan. Sci Total Environ 468-469 (2014): 523-529.