Császár-Nagy N1,2, Bókkon I.2,3,*
1National University of Public Services, H-1083 Budapest, Hungary
2Psychosomatic Outpatient Clinics, H-1037 Budapest, Hungary
3Neuroscience and Consciousness Research Department, Vision Research Institute, Inc. Lowell, MA 01854 USA
*Corresponding Author: István Bókkon, Psychosomatic Outpatient Clinics, H-1037 Budapest, Hungary.
Neuroscience and Consciousness Research Department, Vision Research Institute, Inc.
Lowell, MA 01854 USA
Received: 27 June 2025; Accepted: 04 July 2025; Published: 16 July 2025.
DOI: 10.26502/jppd.2572-519X0251
ShareWe recently suppose that during pregnancy, stress-induced long-term implicit memory (SLEIM) might persist throughout life via the enteric nervous system (ENS) through epigenetic processes, distinct from the central nervous system (CNS). These memories could be pivotal in initiating and sustaining irritable bowel syndrome (IBS). We later improved on our original hypothesis. Specifically, prenatal stressors can alter the mother’s gut microbiota by perturbing the Hypothalamic-Pituitary-Adrenal (HPA) axis and elevating cortisol levels in the blood. Due to the ability of maternal cortisol to bypass the placental barrier, the fetus’s cortisol levels can also increase, which disrupts the HPA axis, affecting the fetus’s intestinal permeability, microbial metabolites, and gut microbiota. Epigenetic modifications induced by microbial metabolites such as short-chain fatty acids – which also influence the development and epigenetic processes of the fetal ENS – may lead to various gut-related illnesses. The bidirectional microbiota-gut-brain axis (MGBA) may convey this SLEIM information from the embryonic ENS to the CNS, resulting in the malfunctioning of emotional and pain processes. Here we assume that the fetal ENS records SLEIM during pregnancy as primordial emotional information and conveys it to the CNS via MGBA. However, fetal CNS cannot interpret these stress signals from the ENS since these are not linked to representations and can operate autonomously of the CNS. The CNS tries to manage the SLEIM signals, triggering stress response systems such as the HPA axis and the immune system, which in turn influence intestinal processes.
Irritable bowel syndrome, Enteric nervous system, Epigenetic implicit memory, Primordial emotions.
Irritable bowel syndrome articles; Enteric nervous system articles; Epigenetic implicit memory articles; Primordial emotions articles.
1. Introduction
The enteric nervous system (ENS) serves a fundamental function as an integrating hub for controlling gastrointestinal physiology [1]. Epigenetic regulation plays a crucial role in the functioning of the ENS [2-5]. The ENS can also operate independently of any neural input from the CNS [6]. In addition, evidence suggests that the ENS may possess the capacity for learning and memory [7-9]. According to Schemann et al. [9], the ENS functions similarly to a “little brain” or “second brain” within the gut, capable of memorization and implicit learning. According to a new assumption we recently presented [10], psychological stressors such as maternal depression and anxiety may induce stress-induced long-term epigenetic implicit memory (SLEIM) in the fetal ENS. These stress-associated memories may persist throughout an individual’s lifetime in the fetal ENS. SLEIM in the fetal ENS can subtly disrupt intestinal homeostasis processes, which can then relay these signals to the brain. These signals might result in symptoms such as bloating, stomach pain, and changes in bowel habits like diarrhea, constipation, or both. However, they may not be potent enough to cause organic changes. Additionally, the microbiota-gut-brain axis (MGBA) serves as a pathway through which SLEIM from the ENS can infiltrate the central nervous system (CNS), impeding the CNS’s ability to process pain and emotional responses [11-15]. Consequently, disturbed cortical-limbic pathways may induce negative emotions and increase perceived pain sensitivity (somatization).
Even though the CNS has taken steps to mitigate the effects of the SLEIM signals, the MGBA continues to transmit these signals from the ENS to the CNS. However, it is the responsibility of the CNS to manage the inflow of SLEIM signals from the gut, which in turn influences intestinal function by initiating stress response systems such as the Hypothalamic-Pituitary-Adrenal (HPA) axis and the immune system. Consequently, a vicious cycle arises between the ENS and the CNS.
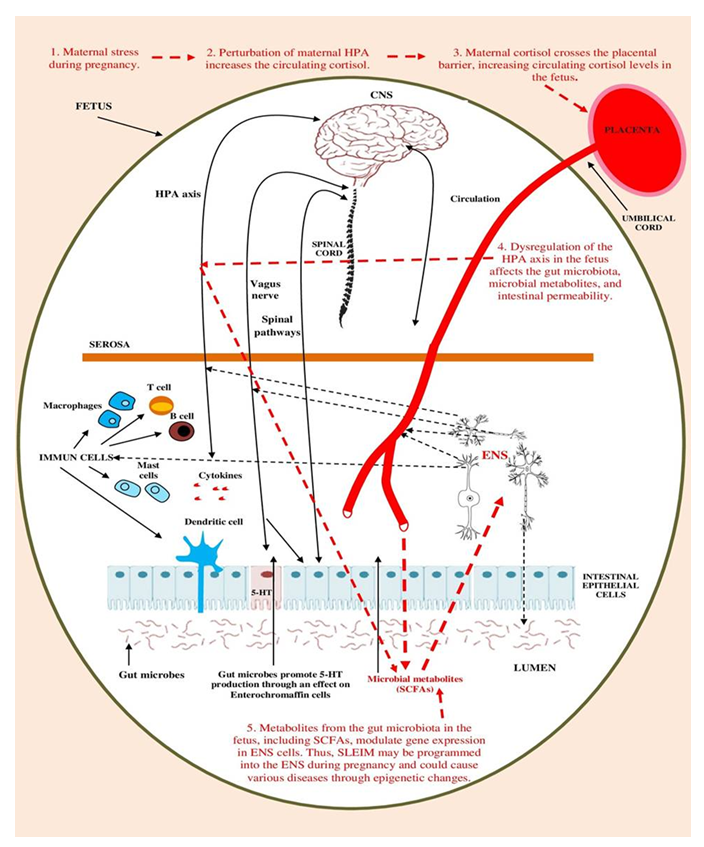
We later elaborated on our initial hypothesis and pointed out the potential of maternal stresses in producing SLEIM in the fetal ENS (Figure 1) [16]. When maternal prenatal stress occurs, it results in an imbalance of the HPA axis that escalates circulating cortisol levels, which could then impact the maternal gut microbiota [17]. This elevated maternal cortisol can permeate the placental barrier, raise the circulating cortisol levels in the fetus, and induce a dysregulation of the HPA axis. This, in turn, may affect the gut microbiota, microbial metabolites, and intestinal permeability, among other things [18]. Moreover, cortisol receptors can be identified in several gut cells, including immune cells, enteroendocrine cells, enteric neurons, and epithelial cells, which suggest that cortisol directly influences gut functionality [19]. The primary metabolites of the microbiota, SCFAs [20], are capable of regulating the cortisol response to psychological stress [21]. These SCFAs can influence multiple mechanisms along the MGBA through direct, and indirect pathways, and epigenetic signaling [22]. Additionally, SCFAs can regulate fetal ENS development [23]. The metabolites of the gut microbiome, notably SCFAs, can affect various disorders by inducing epigenetic alterations through DNA methylation, histone modification, and non-coding RNA-associated gene silencing [24, 22]. The processes mentioned above suggest that SLEIM can be preprogrammed into the fetal ENS during pregnancy. This could endure for a lifetime and significantly contribute to the onset and duration of IBS.
Here we link our previous hypothesis [16] – that fetal ENS may be preprogrammed with implicit, long-term epigenetic memories related to stress experienced during pregnancy – to the new theory suggested by Boem et al. [25]. This theory concerns embodied cognition and perception towards the gut complex, explicitly, the primordial emotions (proto-cognition) in the ENS. We suggest that fetal ENS is able to record implicit, long-term epigenetic stress memories during pregnancy as primordial and non-representational emotional information [16, 25]. This may clarify why it is challenging to access the SLEIM of ENS and treat IBS using various methods and medications.
Figure 1: The parts and lines marked in red represent our simplified hypothesis about how stress-induced long-term epigenetic memory (SLEIM) may be programmed into the fetal ENS during pregnancy. The other processes in Figure 1 indicate complex processes of the intestine that could also play various roles in these processes. This figure is adapted from our open access paper: Császár-N N, Bob P, Bókkon I. Long-Term Implicit Epigenetic Stress Information in the Enteric Nervous System and its Contribution to Developing and Perpetuating IBS. Curr Neuropharmacol. 2024;22(13):2100-2112. https://pmc.ncbi.nlm.nih.gov/articles/PMC11337685/
2. Irritable Bowel Syndrome: Comorbid psychiatric illnesses are present in 50–90% of ibs patients
Between 9% and 23% of people worldwide suffer from irritable bowel syndrome (IBS), a persistent and debilitating Functional Gastrointestinal Disorder (FGID) [26, 27]. IBS is a multi-factorial and heterogeneous disorder that engages pathophysiological central, peripheral, and genetic factors [28]. Common symptoms of IBS include frequent stomach pain, bloating, and alterations in bowel habits such as diarrhea, constipation, or alternating bouts of constipation and diarrhea, all of which occur without detectable structural and biochemical abnormalities [29]. Patients with IBS also frequently experience digestive symptoms such as nausea, non-cardiac chest pain, dyspepsia, and dysphagia [30]. Women are more frequently affected by IBS than men; however, the reasons for this gender prevalence remain unclear. Furthermore, compared to men with IBS, women have been found to experience higher levels of fatigue, anxiety, sadness, and a diminished quality of life [31].
The possible mechanisms for the pathophysiology of IBS have been linked to multiple factors. These include genetic and epigenetic factors, impaired immune processes, stress-related effects on the nervous and endocrine systems, HPA axis dysregulation, disorders in the interaction of the brain-gut axis, dysbiosis, altered gastrointestinal motility, visceral hypersensitivity, infections, food sensitivity, carbohydrate malabsorption, and abnormalities in serotonin metabolism. However, the exact cause of the condition remains unknown [32, 26, 33].
Psychiatric and mood disorders, including anxiety, depression, bipolar disorder, suicidal thoughts, and eating disorders, are increasingly implicated in the development and maintenance of IBS [34-38]. It appears that IBS is the most prevalent disorder related to bidirectional communication via the MGBA [28]. IBS may be a condition characterized by disruptions in brain-gut interactions and stress response systems, such as the autonomic nervous system (ANS) and HPA axis, with corticotropin-releasing hormone-dependent dysregulation of the MGBA implicated in its presentation [39].
Comorbid psychiatric illnesses are present in 50-90% of IBS patients, with anxiety disorders and depression being the most common [40, 41]. IBS patients are three times more likely to suffer from anxiety or depression than healthy individuals [42]. Additionally, studies have revealed familial clusters of IBS, suggesting a potential generational inheritance aspect [43-45]. While numerous factors may contribute to the development of IBS, scientific evidence suggests that stress could be a key factor in its onset. Psychotherapy, medication, dietary changes, microbiota transplantation, and complementary and alternative therapies are some of the current methods used to treat IBS [46-48]. We should mention that the Vagus nerve stimulation could be a promising treatment method for intestinal abnormalities and depressive symptoms in IBS [49, 50]. It’s important to remember that these therapies are intended to IBS alleviate symptoms rather than provide a complete cure for IBS.
3. Maternal stress and anxiety during pregnancy
Maternal stress during pregnancy significantly impacts fetal development and leaves a lasting effect on various offspring’s developmental areas such as metabolic functioning, cognitive growth, and emotional maturation [51, 52]. Pregnancy may be the most mentally sensitive period in a woman’s life. Approximately 10% of pregnant women experience symptoms that meet the criteria for a major depressive disorder, and up to 20% may face depressed symptoms during their pregnancy [53]. Other studies have reported that up to 25% of pregnant women experience high levels of stress, which is associated with a higher risk of unfavorable delivery outcomes and poor mental health postpartum [54, 55]. Moreover, maternal exposure to traumatic events during pregnancy has been linked to a higher lifetime risk of psychiatric problems [56]. Furthermore, the fear of childbirth experienced by women before a first pregnancy mirrors the fears reported by those who are currently pregnant [57]. One in five women suffer from peripartum anxiety disorders, which are more prevalent than previously assumed [58]. However, [59] emphasize the pivotal and deleterious role of maternal stress during pregnancy. The link between the intestinal microbiota and the HPA system is the primary communication mechanism within the microbiota gut-brain axis (MBGA) [60]. Maternal cortisol has garnered significant interest due to its critical role in the physiology of fetal growth and maturation [61]. The wellbeing of both mother and fetus can be negatively affected by disruptions in the operation of the maternal and fetal HPA systems, often resulting from maternal stress [62]. Maternal cortisol can disturb gut microbiota, microbial metabolites, intestinal permeability, and other processes. Given that it can cross the placental barrier, it can increase circulating levels in the fetus and result in dysregulation of the HPA axis [60].
4. The enteric nervous system with memory and implicit learning
The gastrointestinal tract is the only internal organ that develops its own independent nervous system, known as the ENS. The ENS is the largest and most complex unit of the peripheral nervous system. The gastrointestinal tract (GIT) is innervated by approximately 200–600 million neurons and glial cells embedded within its walls [63]. The ENS plays a crucial role as an integrating hub for controlling gastrointestinal physiology [1]. Enteric neurons and their networks have traditionally been considered persistent throughout life. However, growing evidence suggests that neurogenesis and plasticity exist within the postnatal ENS [64-67]. Most of the neurotransmitters secreted by the ENS are also found in the CNS, such as acetylcholine, GABA, dopamine, and serotonin, among others [68, 69]. The development of the ENS’s millions of neurons and glial cells from neural crest cells (NCCs) is governed by genetic, epigenetic, and various signaling processes [70]. Proper epigenetic regulation is crucial for the ENS’s functionality [3, 5]. Epigenetic control plays a pivotal role in the ENS’s operation [2-5].
For instance, studies indicate that the pathophysiology of Hirschsprung’s disease can be affected by alterations in gene expression, which include DNA methylation, histone modification, and microRNA regulation, occurring during ENS development [4, 3]. Additionally, microbiota-sensitive epigenetic mechanisms have been detected in local intestinal cells and peripheral tissues [71]. A variety of embryological model systems, such as zebrafish, birds, and rats, have been leveraged to investigate ENS development [72]. Specifically, the zebrafish is considered a crucial model for understanding vertebrate ENS development [73]. [74] discovered in zebrafish studies that the ENS symptoms observed in double mutants for uhrf1 and dnmt1 are no more severe than those in single mutants. They suggested that complete DNA methylation is required for appropriate ENS formation and that Uhrf1 and Dnmt1 function together. Hence, dnmt1 and uhrf1 may be new candidates for Hirschsprung’s disease. The ENS is a complex and autonomous neural system that develops independently and before the CNS. Often referred to as the “first brain” in evolutionary terms, the ENS can operate without any neural input from the CNS even though they can communicate bidirectionally via the MGBA [6, 75]. Moreover, several studies suggest that the ENS might possess the capacity for learning and memory [7-9]. In other words, the ENS could function as a “little brain” within the gut, playing a role in memory retention and implicit learning [9].
5. Primordial emotions encoded in the fetal ENS
Recently, a new concept was suggested related to embodied cognition and perception, specifically in relation to the gut complex [25]. This new theory suggested that embodied cognition is not representational but instead distributes cognition in or through the body without relying on the concept of representation. The primordial emotions are postulated as the evolutionary beginning of consciousness [76]. They frequently signal that the very existence of the organism is threatened [77, 76]. Emotions play a critical role in the evolution of consciousness and the operations of all mental processes [78]. Primitive (or primordial) emotions, such as thirst, hunger for air and food, or pain, provides longer-lasting intentions and motivations, in addition to experiences [76]. Studies on Hydra have provided evolutionary evidence that the ENS developed before the CNS [75]. Despite lacking a brain or spinal cord, Hydra has maintained an innate neural system for over 600 million years. Moreover, even organisms devoid of a nervous system, such as protists, fungi, and plants, can exhibit various forms of elementary learning [79]. Furthermore, physical parts, activities, or content can play a decisive role in forming and processing cognitive processes.
In the gut, unconscious perception is viewed as a principle characteristic of visceral perception, occurring “largely outside of awareness” [80]. Additionally, unconscious mechanisms that dictate behavior include gut microorganisms, which influence fundamental behavioral patterns and cognitive function [81]. Furthermore, epigenetic processes significantly contribute to the development of both unconscious and conscious memory processes [16, 82-85].
Several theories attempt to explain the complex nature of emotions. However, there are over 90 definitions in the literature about emotion [77]. Theories about emotions conventionally divide into two categories. According to cognitive theories, emotions are or essentially involve evaluative thoughts or judgments. The non-cognitive theories claim that an emotion can occur without any thought. Simply put, emotions are reactions that people experience in response to events or situations. Primordial emotions such as thirst, hunger for air, hunger for food, pain, etc. constitute the subjective component of instincts, which are genetically programmed behavioral patterns that result in homeostasis [76]. Developing emotional systems play a key role in the cognitive representational development [86]. We assume that maternal stress generates SLEIMs in the fetal ENS, which actually belong to primordial emotions (general, non-representational fears) that the fetal ENS can record.
6. Conclusions
The ENS can operate independently of the CNS [6]. This indicates that, during embryonic development post-conception, the early-formed ENS (with primordial emotions and very basic cognition) can execute implicit learning independently from the CNS. As previously discussed, a significant proportion of pregnant women endure high stress levels, which potentially disrupts fetal ENS development due to stress-induced dysbiosis. Information regarding this maternal stress, in the form of primordial emotions, may be encoded within the fetal ENS via implicit, enduring epigenetic processes, separate from the CNS. Maternal stress can instigate dysbiosis, disturb gut microbiota and microbial metabolites, and affect intestinal permeability, among other processes. These factors can be influenced by maternal cortisol, which can permeate the placental barrier, elevate circulating levels, and cause HPA axis dysregulation through the MGBA [60]. The pregnant women’s emotional states affect actual fetal development [87]. However, the fetus does not know what stressful events are happening in its mother, it only senses that increased cortisol levels are entering its system through the placenta, which creates a general, unrepresentative fear (primordial emotions) in it. We hypothesize that maternal stress creates SLEIM in the fetal ENS [16], which are primordial emotions that the fetal ENS can record independently of the CNS. In other words, the fetal CNS cannot interpret these implicit primordial stress signals from the ENS since they are not linked to representations and operate independently of the CNS. Despite this, the CNS must deal with the influx of SLEIM signals from the ENS, triggering stress response systems such as the HPA axis and the immune system, which in turn influence intestinal processes. Between the ENS and the CNS, this interplay creates a "vicious circle" resulting in the malfunctioning of emotional and pain processes in the CNS. Our assumption may help to understand why IBS is difficult to treat with various methods and medications. However, it’s crucial to remember that diverse therapies may reduce symptoms rather than offer a full recovery from IBS.
Funding
This work was not supported by any funding.
Author contributors
I.B. developed the concept. N.Cs-N. and I.B. contributed to the
writing of the manuscript.
Declaration of competing interest
The authors report no financial and non-financial conflicts of interest. The authors alone are responsible for all the content.
References
- Bon-Frauches A C, Boesmans W. The enteric nervous system: the hub in a star network. Nat. Rev. Gastroenterol. Hepatol 17 (2020): 717-718.
- Diposarosa R, Bustam N A, Sahiratmadja E, et al. Literature review: enteric nervous system development, genetic and epigenetic regulation in the etiology of Hirschsprung's disease. Heliyon (2021): e07308.
- Torroglosa A, Villalba-Benito L, Toro L B, et al. Epigenetic mechanisms in hirschsprung disease. Int. J. Mol. Sci 20 (2019): 3123.
- Jaroy E G, Acosta-Jimenez L, Hotta R, et al. "Too much guts and not enough brains": (epi)genetic mechanisms and future therapies of Hirschsprung disease - a review. Clin. Epigenetics 11 (2019): 35.
- Cheng L. Progress on the regulation of DNA methylation in the development of the enteric nervous system. Int. J. Pediatr (2018): 756-760.
- Spencer N J, Hu H. Enteric nervous system: sensory transduction, neural circuits and gastrointestinal motility. Nat. Rev. Gastroenterol. Hepatol 17 (2020): 338-351.
- Annahazi A, Schemann M. The enteric nervous system: “A little brain in the gut”. Neuroforum 26 (2020): 31-42.
- Furness J B, Clerc N, Kunze W A. Memory in the enteric nervous system. Gut 47 Suppl 4 (2000): 60-76.
- Schemann M, Frieling T, Enck P. To learn, to remember, to forget-how smart is the gut? Acta Physiol 228 (2020): e13296.
- Császár -N Noémi, Bókkon I. Hypnotherapy and IBS: Implicit, long-term stress memory in the ENS? Heliyon 9 (2022): e12751.
- Nisticò V, Rossi R E, D’Arrigo A M, et al. Functional neuroimaging in irritable bowel syndrome: A systematic review highlights common brain alterations with functional movement disorders. J. Neurogastroenterol. Motil 28 (2022): 185-203.
- Li J, He P, Lu X, et al. A resting-state functional magnetic resonance imaging study of whole-brain functional connectivity of voxel levels in patients with irritable bowel syndrome with depressive symptoms. J. Motil 27 (2021): 248-256.
- Mao C P, Chen F R, Huo J H, et al. Altered resting-state functional connectivity and effective connectivity of the habenula in irritable bowel syndrome: A cross-sectional and machine learning study. Hum. Brain Mapp 41(2020): 3655-3666.
- Bhatt R R, Gupta A, Labus J S, et al. Altered brain structure and functional connectivity and its relation to pain perception in girls with irritable bowel syndrome. Psychosom. Med 81 (2019): 146-154.
- Qi R, Liu C, Weng Y, et al. Disturbed interhemispheric functional connectivity rather than structural connectivity in irritable bowel syndrome. Front. Mol. Neurosci 9 (2016): 141.
- Császár-N N, Bob P, Bókkon I. Long-Term Implicit Epigenetic Stress Information in the Enteric Nervous System and its Contribution to Developing and Perpetuating IBS. Curr. Neuropharmacol 22 (2024): 2100-2112.
- Mepham J, McGee N T, Andrews K, et al. Exploring the effect of prenatal maternal stress on the microbiomes of mothers and infants: A systematic review. Dev. Psychobiol 65 (2023): e22424.
- Gorczyca K, Obuchowska A, Trojnar K Z, et al. Changes in the gut microbiome and pathologies in pregnancy. Int. J. Environ. Res. Public Health 19 (2022): 9961.
- Rusch J A, Layden B T, Dugas L R. Signalling cognition: the gut microbiota and hypothalamic-pituitary-adrenal axis. Front. Endocrinol 14 (2023): 1130689.
- Silva Y P, Bernardi A, Frozza R L. The role of short-chain fatty acids from gut microbiota in gut-brain communication. Front. Endocrinol 11 (2020): 25.
- Dalile B, Vervliet B, Bergonzelli G, et al. Colon-delivered short-chain fatty acids attenuate the cortisol response to psychosocial stress in healthy men: a randomized, placebo-controlled trial. Neuropsychopharmacology 45 (2020): 2257-2266.
- Stein R A, Riber L. Epigenetic effects of short-chain fatty acids from the large intestine on host cells. Microlife 4 (2023): 032.
- Sajdel-Sulkowska E M. The Impact of Maternal Gut Microbiota during Pregnancy on Fetal Gut-Brain Axis Development and Life-Long Health Outcomes. Microorganisms 11 (2023): 2199.
- Lan Z, Tang X, Lu M, et al. The role of short-chain fatty acids in central nervous system diseases: A bibliometric and visualized analysis with future directions. Heliyon 10 (2024): e26377.
- Boem F, Greslehner G P, Konsman J P, et al. Minding the gut: extending embodied cognition and perception to the gut complex. Front. Neurosci.17 (2024): 1172783.
- Saha L. Irritable bowel syndrome: pathogenesis, diagnosis, treatment, and evidence-based medicine. World. J. Gastroenterol 20 (2014): 6759-6773.
- Adriani A, Ribaldone D, Astegiano M, et al. Irritable bowel syndrome: the clinical approach. Panminerva Med 60 (2018).
- Mayer E A, Ryu H J, Bhatt R R. The neurobiology of irritable bowel syndrome. Mol. Psychiatry. 28 (2023): 1451-1465
- Hamarashid B R, Dalkiliç S, Dalkiliç L K, et al. Irritable Bowel Syndrome (IBS): A Review. Biosci 11 (2020): 36-52.
- Chong P P, Chin V K, Looi C Y, et al. The Microbiome and Irritable Bowel Syndrome - A Review on the Pathophysiology, Current Research and Future Therapy. Front. Microbiol 10 (2019): 1136.
- Kim Y S, Kim N. Sex-Gender Differences in Irritable Bowel Syndrome. J. Neurogastroenterol. Motil 24 (2018): 544-558.
- Holtmann G J, Ford A C, Talley N J. Pathophysiology of irritable bowel syndrome. The Lancet Gastroenterol. Hepatol 1 (2016): 133-146.
- Camilleri M, Lasch K, Zhou W. Irritable bowel syndrome: methods, mechanisms, and pathophysiology. The confluence of increased permeability, inflammation, and pain in irritable bowel syndrome. Am. J. Physiol. Gastrointest. Liver Physiol 303 (2012): 775-785.
- Tarar Z I, Farooq U, Zafar Y, et al. Burden of anxiety and depression among hospitalized patients with irritable bowel syndrome: a nationwide analysis. Ir. J. Med. Sci 192 (2023a): 2159-2166.
- Tarar M, Vasupanrajit A, Jirakran K, et al. Adverse childhood experiences and reoccurrence of illness impact the gut microbiome, which affects suicidal behaviours and the phenome of major depression: towards enterotypic phenotypes. Acta Neuropsychiatr 13 (2023b): 1-18.
- Zhu F, Tu H, Chen T. The Microbiota-Gut-Brain Axis in Depression: The Potential Pathophysiological Mechanisms and Microbiota Combined Antidepression Effect. Nutrients 14 (2022): 2081.
- Clapp M, Aurora N, Herrera L, et al. Gut microbiota's effect on mental health: The gut-brain axis. Clin. Pract 7 (2017): 987.
- Lydiard R B. Irritable bowel syndrome, anxiety, and depression: what are the links? J. Clin. Psychiatry 62 (2001): 38-45.
- Kano M, Muratsubaki T, Van Oudenhove L, et al. Altered brain and gut responses to corticotropin-releasing hormone (CRH) in patients with irritable bowel syndrome. Sci. Rep 7 (2017): 12425.
- Chhiber S. Psychiatric comorbidity in patients of irritable bowel syndrome. Indian J. Psychiatry 64 (2022): S688.
- Banerjee A, Sarkhel S, Sarkar R, et al. Anxiety and Depression in Irritable Bowel Syndrome. Indian J. Psychol. Med 39 (2017): 741-745.
- Klem F, Wadhwa A, Prokop L J, et al. Prevalence, Risk Factors, and Outcomes of Irritable Bowel Syndrome After Infectious Enteritis: A Systematic Review and Meta-analysis. Gastroenterology 152 (2017): 1042-1054.
- Makker J, Chilimuri S, Bella J N. Genetic epidemiology of irritable bowel syndrome. World J. Gastroenterol 21 (2015): 11353-11361.
- Saito Y A, Petersen G M, Larson J J, et al. Familial aggregation of irritable bowel syndrome: a family case-control study. Am. J. Gastroenterol 105 (2010): 833-841.
- Saito Y A, Zimmerman J M, Harmsen W S, et al. Irritable bowel syndrome aggregates strongly in families: a family-based case-control study. Neurogastroenterol. Motil 20 (2008): 790-797.
- Papale A J, Flattau R, Vithlani N, et al. A Review of Pharmacologic and Non-Pharmacologic Therapies in the Management of Irritable Bowel Syndrome: Current Recommendations and Evidence. J. Clin. Med 13 (2024): 6948.
- Tetali B, Suresh S. Management of irritable bowel syndrome: a narrative review. Transl. Gastroenterol. Hepatol 9 (2024): 26.
- Tripathi R, Mehrotra S. Irritable bowel syndrome and its psychological management. Ind. Psychiatry. J 24 (2015): 91-93.
- Veldman F, Hawinkels K, Keszthelyi D. Efficacy of vagus nerve stimulation in gastrointestinal disorders: a systematic review. Gastroenterol. Rep. (Oxf) 13 (2025): 009.
- Yan Q, Chen J, Ren X, et al. Vagus Nerve Stimulation Relives Irritable Bowel Syndrome and the Associated Depression via α7nAChR-mediated Anti-inflammatory Pathway. Neuroscience 530 (2023): 26-37.
- Van den Bergh B, van den Heuvel M I, Lahti M, et al. Prenatal developmental origins of behavior and mental health: The influence of maternal stress in pregnancy. Neurosci. Biobehav. Rev 117 (2020): 26-64.
- Beijers R, Buitelaar J K, Weerth C D. Mechanisms underlying the effects of prenatal psychosocial stress on child outcomes: beyond the HPA axis. Eur. Child Adolesc. Psychiatry 23 (2014): 943-956.
- Kinser P A, Thacker L R, Lapato D, et al. Depressive Symptom Prevalence and Predictors in the First Half of Pregnancy. J. Womens Health 27 (2018): 369-376.
- Ibrahim S M, Lobel M. Conceptualization, measurement, and effects of pregnancy-specific stress: Review of research using the original and revised Prenatal Distress Questionnaire. J. Behav. Med. Brain Sci 43 (2020): 16-33.
- Mahaffey B L, Lobel M. Pregnancy. In: Handbook of Health Psychology. New York, NY: Routledge (2018): 450-461.
- Talge N M, Neal C, Glover V. Early Stress, Translational Research and Prevention Science Network: Fetal and Neonatal Experience on Child and Adolescent Mental Health. Antenatal maternal stress and long-term effects on child neurodevelopment: how and why? J. Child Psychol. Psychiatry 48 (2007): 245-261.
- Rondung E, Magnusson S, Ternström E. Preconception fear of childbirth: experiences and needs of women fearing childbirth before first pregnancy. Reprod. Health 19 (2022): 202.
- Fawcett E J, Fairbrother N, Cox M L, et al. The Prevalence of Anxiety Disorders During Pregnancy and the Postpartum Period: A Multivariate Bayesian Meta-Analysis. J. Clin. Psychiatry 80 (2019): 18r12527.
- Zietlow A L, Nonnenmacher N, Reck C, et al. Emotional Stress During Pregnancy - Associations with Maternal Anxiety Disorders, Infant Cortisol Reactivity, and Mother-Child Interaction at Pre-school Age. Front. Psychol 10 (2019): 2179.
- Turroni F, Rizzo S M, Ventura M, et al. Cross-talk between the infant/maternal gut microbiota and the endocrine system: A promising topic of research. Microbiome Res. Rep 1 (2022): 14.
- Benediktsson R, Calder A A, Edwards C R, et al. Placental 11 beta-hydroxysteroid dehydrogenase: a key regulator of fetal glucocorticoid exposure. Clin. Endocrinol 46 (1997): 161-166.
- Tarlan A, Sáinz F, Riggat A. Relaciones, Vivencias y Psicopatología, 1st ed.; Herder Editorial: Barcelona, Spain (2013).
- Geng Z H, Zhu Y, Li Q L, et al. Enteric Nervous System: The Bridge Between the Gut Microbiota and Neurological Disorders. Front. Aging Neurosci 14 (2022): 810483.
- Bornstein J C. Experimental West Nile virus infection provides lessons for recovery from enteric neuropathies. J. Clin. Invest 134 (2024): e185865.
- Stavely R, Rahman A A, Mueller J L, et al. Mature enteric neurons have the capacity to reinnervate the intestine with glial cells as their guide. Neuron 112 (2024): 3143-3160.
- Kulkarni S, Micci M A, Leser J, et al. Adult enteric nervous system in health is maintained by a dynamic balance between neuronal apoptosis and neurogenesis. Proc. Natl. Acad. Sci. U S A 114 (2017): 3709-3718.
- Gershon M D. Behind an enteric neuron there may lie a glial cell. J. Clin. Invest 121 (2011): 3386-3389.
- Nezami B G, Srinivasan S. Enteric nervous system in the small intestine: pathophysiology and clinical implications. Curr. Gastroenterol. Rep 12 (2010): 358-365.
- Krantis A. GABA in the Mammalian Enteric Nervous System. News Physiol. Sci 15 (2000): 284-290.
- Uribe R A. Genetic regulation of enteric nervous system development in zebrafish. Biochem. Soc. Trans 52 (2024): 177-190.
- Woo V, Alenghat T. Epigenetic regulation by gut microbiota. Gut Microbes 4 (2022): 2022407.
- Heanue T A, Shepherd I T, Burns A J. Enteric nervous system development in avian and zebrafish models. Dev. Biol 417 (2016): 129-138.
- Kuil L E, Chauhan RK, Cheng W W, et al. Zebrafish: A model organism for studying enteric nervous system development and disease. Front. Cell. Dev. Biol 8 (2021): 629073.
- Ganz J, Melancon E, Wilson C, et al. Epigenetic factors Dnmt1 and Uhrf1 coordinate intestinal development. Dev. Biol 455 (2019): 473-484.
- Furness J B, Stebbing M J. The first brain: species comparisons and evolutionary implications for the enteric and central nervous systems. Neurogastroenterol. Motil 30 (2018): e13234.
- Denton D A, McKinley M J, Farrell M, et al. The role of primordial emotions in the evolutionary origin of consciousness. Conscious Cogn 18 (2009): 500-514.
- Denton D A. The primordial emotions: The dawning of consciousness. Oxford University Press (2006).
- Izard C E. Emotion theory and research: highlights, unanswered questions, and emerging issues. Annu. Rev. Psychol 60 (2009): 1-25.
- van Duijn M. Phylogenetic origins of biological cognition: convergent patterns in the early evolution of learning. Interface Focus 7 (2017): 20160158.
- Ádám G. Visceral perception. Boston, MA: Springer US (1998): 87-12.
- Dinan T G, Stilling R M, Stanton C, et al. Collective unconscious: how gut microbes shape human behavior. J. Psychiatr. Res 63 (2015): 1-9.
- Liester M B, Sullivan E E. A review of epigenetics in human consciousness. Cogent Psychology 6 (2019): 1668222.
- Grigorenko E L, Kornilov S A, Naumova O Y. Epigenetic regulation of cognition: A circumscribed review of the field. Dev. Psychopathol 28 (2016):1285-1304.
- Rosales-Reynoso M A, Ochoa-Hernández A B, Juárez-Vázquez C I., et al. Epigenetic mechanisms in the development of memory and their involvement in certain neurological diseases. Neurologia 31 (2016): 628-638.
- Bókkon I, Vas J P, Császár N, et al. Challenges to free will: transgenerational epigenetic information, unconscious processes, and vanishing twin syndrome. Rev. Neurosci 25 (2014): 163-175.
- Toronchuk JA, Ellis GF. Affective neuronal selection: the nature of primordial emotion systems. Front. Psychol 3 (2013): 589.
- Kim D, Lee I, Bang K S, et al. Do the Emotions of Middle-Income Mothers Affect Fetal Development More Than Those of High-Income Mothers? - The Association between Maternal Emotion and Fetal Development. Int. J. Environ. Res. Public Health 16 (2019): 2065.