Sergio Eduardo Tricta Quaresma, Guilherme Veiga Guimaraes, Ricardo Simões Neves, Maria Cristina Marino de Oliveira, Itamara Lucia Itagiba Neves, Cesar José Gruppi, Edimar Alcide Bocchi*
Heart Institute, University of São Paulo School of Medicine Hospital das Clínicas, São Paulo, Brazil
*Corresponding author: Edimar Alcides Bocchi, Heart Institute, University of São Paulo School of Medicine Hospital das Clínicas, Dr Eneas de Carvalho Aguiar Avenue, CEP 05403-000, São Paulo, Brazil
Received: 02 October 2019; Accepted: 15 October 2019; Published: 22 April 2020
Objectives: The use of a local anesthesia solution with vasoconstrictor for the dental procedure of patients with heart disease is controversial since it could generate adverse cardiovascular risk. Also, its safety has not been investigated in heart failure (HF) patients. This study sought to determine the efficacy and safety of a vasoconstrictor in dental interventions in heart failure (HF) patients with left ventricular reduced ejection fraction (HFrEF) and optimized treatment.
Study Design: prospective, parallel group, controlled, double-blind study (Tooth HF study). Materials and Methods: HF patients with ejection fraction <45% and with optimized therapy were randomized to dental intervention using anesthesia solution lidocaine without epinephrine (LSE) or anesthetic solution lidocaine with epinephrine (LCE). The primary endpoint was pain during the intervention.
Results: Seventy-two patients were randomized to LSE (n=36) or LCE (n=36). A high prevalence of poor oral health was observed. Reduced pain was observed in LCE patients during dental extraction but not during dental restorations. Ten LSE patients had pain versus 4 LCE patients (p=0.037). No differences between the LSE and LCE patients were observed concerning 24-hour monitored systemic blood pressure, heart rate, and arrhythmia. However, systemic blood pressure increased, and heart rate significantly decreased in relation to the baseline phase during and after the procedure in both groups.
Conclusion: Association of local anesthesia with a vasoconstrictor was more effective for pain control in HFrEF patients without compromising safety. These results will benefit millions of HFrEF patients around the world in need of dental intervention.
Dental Procedure; Heart Failure; Lidocaine; Epinephrine; Anesthesia
Dental Procedure articles, Heart Failure articles, Lidocaine articles, Epinephrine articles, Anesthesia articles
Abbreviations: CHD-coronary heart disease; CHF-coronary heart failure; CVD-cardiovascular disease; DMFt- decayed, lost, and filled teeth; ECG-electrocardiogram; HF-heart failure; HfrEF-HF with left ventricular reduced ejection fraction; LVEF- left ventricular ejection fraction; NRS-Numeric Rating Scale
1. Introduction
Poor oral health is associated with cardiovascular (CVD) diseases [1, 2]. The number of teeth declines in a dose-dependent manner in all-cause mortality and mortality from CVD and coronary heart disease (CHD) [3]. A high prevalence of periodontal disease has been reported in CHD [4]. Poor oral health, gingivitis, moderate periodontitis, and severe periodontitis have also been reported in HF patients [5]. Severe periodontitis is more prevalent among chronic HF patients than in the general population. Periodontitis and HF share risk factors, such as smoking, diabetes mellitus, alcohol consumption, hypertension, and low socioeconomic status [5, 6]. Thus, the global burden of poor oral heath in HF is likely to be large and severe. Based on NHAMES 2001 to 2014, it is estimated that 6.5 million Americans > 20 years of age had HF (2.5%) [7]. Projections show that the prevalence of HF will increase to 46% from 2012 to 2030 [7]. Because of progressive heart failure and sudden death due to arrhythmias, HF is associated with substantial mortality despite improvement in treatment.
HF patients with poor oral health frequently need dental interventions with anesthesia. History of HF is a risk factor for significant morbidity and mortality in the perioperative period of interventions [8]. Dental interventions for pain and fear may potentially lead to endogenous catecholamine release, hemodynamic changes, arrhythmias, and additional risk to the HF population. Otherwise, in oral interventions the association of a vasoconstrictor with anesthetic drugs may have the advantage of anesthesia reabsorption inhibition, longer duration, and a deeper effect, reducing bleeding and procedure time. However, vasoconstrictors may potentially initiate hemodynamic disturbance, heart rate changes, and arrhythmias. Although vasoconstrictors are well tolerated by healthy people, their use in heart failure patients is still controversial; therefore, it must be better elucidated. In this double-blind, controlled, prospective investigation, we tested the hypothesis that a local anesthesia plus epinephrine is superior for reducing pain without causing undesirable hemodynamic and arrhythmogenic events in HF patients undergoing dental interventions.
2. Methods
2.1 Study population
Seventy-two male and female patients with ischemic and nonischemic chronic heart failure, at least a 6-year history of HF, left ventricular ejection fraction ≤ 45%, New York Heart Association functional class II and III/IV, and drug optimized therapy for the previous 6 months were included. Baseline characteristics are reported in Table 1. Patients were excluded in case of recent myocardial infarction, complex arrhythmia, uncontrolled hypertension, pregnancy, neoplasms, allergy to lidocaine and epinephrine, antiarrhythmic drugs, and decompensated heart failure.
2.2 Study design
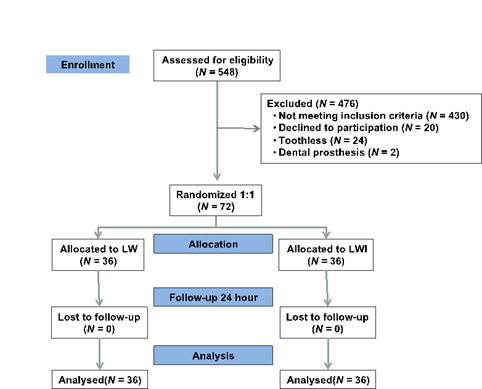
This was a longitudinal, prospective, randomized double-blind study performed at one tertiary cardiology center from December 2014 to September 2017. All patients were recruited from the same center. All referred patients initially had a dental appointment to evaluate oral health and which type of dental procedure might be necessary (Figure 1). Patients who fulfilled the inclusion criteria were invited to participate. After accepting the invitation and signing the consent form, all volunteers were randomized and allocated into two groups: lidocaine plus epinephrine (LCE) and lidocaine without epinephrine (LSE). A computer-generated-random number list program generated the block randomization list characterized by two interventions and two controls. Tooth extraction or restoration was planned as needed. Based on expected greater pain during tooth extraction compared with stratified tooth restoration, randomization was made by using block randomization separately for extraction and tooth restoration to achieve a balance. We stratified according to functional class. The study periods were standardized for both groups: T0 (baseline, one hour before procedure); T1 (injection of local anesthesia); T2 (procedure period); and T3 (after procedure up to one hour). The study was approved by the Institutional Ethics Committee (3820/12/076) and registered at clinicaltrials.gov (NCT022228083).
|
Variable |
Total Group N=72 |
Lidocaine without epinephrine n=36 |
Lidocaine with epinephrine n=36 |
P1 |
|
Age (yrs) |
51 ± 11 |
50 ± 10 |
51 ± 11 |
ns |
|
Male sex, (%) |
45 (62, 5%) |
23 (63, 9%) |
22 (61, 1%) |
ns |
|
Race:white/black/mullato/yellow, % |
79/13/6/3 |
81/14/3/3 |
78/11/8/3 |
ns |
|
Body mass index, kg/m2 |
26 ± 5 |
26 ± 5 |
26 ± 5 |
ns |
|
Etiology, (%) |
ns |
|||
|
Ischemic/chagasic/valvar/IDC |
28/8/3/28 |
25/6/0/31 |
31/11/6/25 |
- |
|
SH/peri-partum/alcoholic/other |
14/8/8/8 |
19/6/17/8 |
8/11/0/8 |
- |
|
NYHA FC II/III/IV, (%) |
42/56/3 |
36/58/6 |
47/53/0 |
ns |
|
LVEF, (%) |
27 ± 8 |
29 ± 8 |
28 ± 7 |
ns |
|
LVEDD, (mm) |
68 ± 10 |
68 ± 9 |
68 ± 10 |
ns |
|
Devices (CRT, ICD), (%) |
14 |
17 |
12 |
ns |
|
Comorbidities, (%) |
ns |
|||
|
Diabetes |
28 |
22 |
33 |
|
|
COPD |
1 |
0 |
2.8 |
|
|
Smoking |
42 |
50 |
33 |
|
|
Hypothyroidism |
10 |
8 |
11 |
|
|
Hyperthyroidism |
3 |
3 |
3 |
|
|
Dyslipidemia |
24 |
28 |
20 |
|
|
Previous cerebral stroke |
8 |
3 |
14 |
|
|
Rhythm, (%) |
ns |
|||
|
Sinus rhythm |
71 |
67 |
75 |
|
|
Atrial fibrillation |
11 |
17 |
6 |
|
|
Pacemaker and sinus rhythm |
8 |
11 |
6 |
|
|
Pacemaker and atrial fibrillation |
3 |
0 |
6 |
|
|
Other |
7 |
6 |
8 |
|
|
HF Pharmacotherapy, (%) |
ns |
|||
|
Beta-blocker |
99 |
100 |
97 |
|
|
ACEI |
67 |
69 |
64 |
|
|
ARB |
26 |
25 |
28 |
|
|
Diuretics |
90 |
86 |
94 |
|
|
Antiplatelet |
38 |
39 |
36 |
|
|
Oral anticoagulant |
31 |
33 |
28 |
|
|
Digital |
38 |
31 |
44 |
|
|
Vasodilator |
33 |
36 |
31 |
|
|
Calcium channel blockers |
6 |
8 |
3 |
|
|
Drugs for hyperlipidemia |
51 |
58 |
44 |
|
|
Oral hypoglycemic |
29 |
28 |
31 |
|
|
Thyroid hormone |
13 |
8 |
17 |
|
|
Drugs for depression |
15 |
17 |
14 |
|
|
Drugs for anxiety |
7 |
6 |
6 |
|
|
Drugs for stomach diseases |
42 |
47 |
36 |
|
IDC=idiopathic dilated cardiomyopathy; SH= systemic hypertension; NYHA FC= New York Heart Association functional class; LVEF= left ventricular ejection fraction; CRT= cardiac resynchronization therapy; ICD= implanted cardiac defibrillator; COPD=chronic obstructive pulmonary disease; HF=heart failure; ACEI=angiotensin converting enzyme inhibitor; ARB=angiotensin 1 receptor blocker.
Table 1: Baseline Patient Characteristics.
2.3 Dental procedure
Group LCE received 2% lidocaine plus epinephrine 1:100, 000 and group LSE 2% lidocaine for anesthesia performed immediately before the dental intervention. Anesthesia was induced carefully with aspiration and slow injection. A minimum of 5 minutes was allowed to obtain anesthesia effectiveness. In both groups, patients received for tooth extraction a standard dose of 2 cartridges (3.6 ml) and for tooth restoration 1 cartridge (1.8 ml) of 2% lidocaine. In case of pain during the procedure, despite the previous anesthesia, patients could receive an additional cartridge, as required. Cartridges were covered with a thin piece of aluminum foil so either patient or dentist could not see the type of anesthetic with or without epinephrine. It was planned that different dentists treated the patients. Sutures were performed after tooth extraction. In case of tooth extraction, if a tooth was considered a source of infection after clinical and radiological evaluation, a prescription of 500 mg of amoxicillin every 8 hours for 7 days or 300 mg of clindamycin to those allergic individuals was provided. To high- or moderate-risk patients for bacterial endocarditis, the Brazilian Cardiology Society Guidelines were followed, and a prescription of 2 g of amoxicillin was provided one hour before the procedure. All patients included in the trial were instructed not to stop with any routine medication, including anticoagulant and antiplatelet agents.
2.4 Pain evaluation during intervention
Patients were instructed to inform the dentist of the exact moment that they felt pain during the dental procedure. If the patient felt pain, the dentist interrupted the intervention and requested the patient to rate the intensity of pain on the Numeric Rating Scale (NRS). If this occurred and the procedure was not over yet, the patient would receive an extra complete cartridge of anesthetic.
2.5 Continuous ambulatory electrocardiographic monitoring (holter)
Twenty-four-hour ECG monitoring was performed using a Holter monitor model Seer Light (GE). The monitor was placed 2 hours before the dental procedure. Data from this monitoring, including heart rate and arrhythmia, were analyzed from period T0 to period T3. Holter monitoring methods and analyses have been published previously [7, 8, 9, 10].
2.6 Ambulatory monitoring of 24-hour blood pressure
After placing a Holter monitor, patients’ blood pressure was monitored for 24 hours. Measurements started immediately. The monitor used in the trial was a Spacelabs ABP model 90207 (Spacelabs Medical Inc. Redmond, WA). Data were averaged for 24 hours, day and night periods, and for systolic and diastolic blood pressure separately. The periods T0 to T3 were analyzed. This analysis method has also been published previously [7, 8, 9, 10].
2.7 Statistical analyses
Sample size was based partly on previous investigation results that analyzed effects of the association of vasoconstrictor and lidocaine (28 pts) versus lidocaine (31 pts) in dental interventions in patients with valvular diseases. Patients in the vasoconstrictor group had fewer complaints (p=0.03), but that finding did not reflect the effect size, the power of the study, or event rate in the population [9]. The sample size used in part was also exploratory because a systematic review did not find reported effects of the association of vasoconstrictor and lidocaine on pain during dental intervention. Data are shown as average ± standard deviation or median and interquartile range. Statistical software SPSS 17.0 for Windows (SPSS inc, Chicago, IL) was used to perform statistical analyses. The Shapiro-Wilk test was used to check normality of the studied population. Fisher’s exact test was used to evaluate homogeneity of the sample in both groups in baseline. ANOVA was used to compare groups according to the periods. The pos-hoc Bonferroni test was used to identify significant differences. Mann-Whitney and Wilcoxon tests were used to identify differences between groups and inside each group, for parametric and nonparametric parameters. The unpaired t test was used to identify differences in the groups in the changes in hemodynamic parameters. Outcomes were considered significant whenever their descriptive levels were lower than 0.05 (p < 0.05)
3. Results
The baseline characteristics of patients in both LSE and LCE groups were comparable (Table 1) except for alcoholic HF etiology. Most patients had an ischemic or idiopathic dilated etiology, white race, male, in functional class II and III, important reduction in LVEF, and all were under optimized treatment for HF. Thirty-four HF patients underwent tooth extraction and 38 patients tooth restoration. No difference was found in the duration of dental procedures between groups (LSE 30 ± 10 mi; LCE 29 ± 16 min, p= 0.34).
3.1 Oral heath in hf (Table 2)
The minority of patients had good oral health. A high percentage was observed of lost teeth, gingivitis, dental calculus, and periodontal disease with high decay, lost and filled teeth.
3.2 Primary endpoint (Table 3)
Lidocaine with epinephrine was associated with a statistically significant reduction in the number of patients who reported pain during tooth extraction but not during tooth restoration. A tendency toward statistical significance in reduction of the NRS pain scale was observed in the group undergoing tooth extraction.
3.3 Safety endpoints (Table 4)
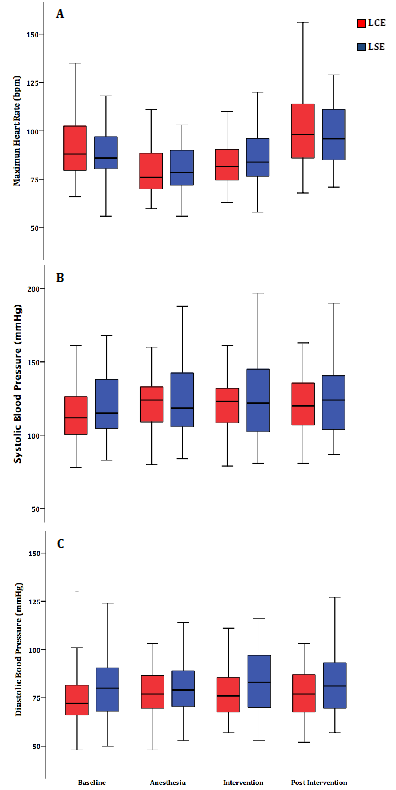
Ventricular extra-systoles, atrial extra-systoles, non-sustained ventricular tachycardias, and supraventricular tachycardias were observed in the baseline period. However, arrhythmias per minute did not increase during the anesthesia, intervention, or post intervention period. Syncope was also reported. In both LSE and LCE groups, the baseline mean systolic and diastolic systemic blood pressures were lower in the baseline period compared with other periods. However, no differences were observed between the LSE and LCE groups. Differences concerning mean heart rate were not observed in the study periods in either group. Also, the mean heart rate was comparable between both groups (Figure 2).
|
Findings |
Total group N=72 |
Lidocaine without epinephrine (n=36) |
Lidocaine with epinephrine (n=36) |
p |
|
Decayed teeth |
3.79 ± 3.66 |
4 ± 3.61 |
3.56 ± 3.74 |
ns |
|
Lost teeth |
10.9 ± 8.64 |
9.58 ± 7.71 |
12.26 ± 9.43 |
ns |
|
Filled teeth |
4.90 ± 4.56 |
4.72 ± 4.23 |
5.09 ± 4.94 |
ns |
|
DMFt index |
19.39 ± 7.18 |
18.81 ± 6.61 |
19.97 ± 7.75 |
ns |
|
Loe and Silness Index |
0.83 ± 0.63 |
0.89 ± 0.69 |
0.77 ± 0.56 |
ns |
|
Gingivitis (%) |
55 (76.4) |
29 (80.6) |
26 (72.2) |
ns |
|
Periodontal disease (%) |
41 (57.7) |
25 (69.4) |
16 (45.7) |
.043 |
|
Dental calculus (%) |
44 (62.0) |
23 (65.7) |
21 (58.3) |
ns |
|
Oral health, n (%) |
ns |
|||
|
Good |
11 (15.3) |
6 (16.7) |
5 (13.9) |
|
|
Reasonable |
37 (51.4) |
17 (47.2) |
20 (55.6) |
|
|
Poor |
24 (33.3) |
13 (36.1) |
11 (30.6) |
|
DMFt= decayed, lost, and filled teeth.
Table 2: Oral Heath Results.
|
Outcome |
Total Group N=72 |
Lidocaine without epinephrine (n=36) |
Lidocaine with epinephrine (n=36) |
p |
|
Tooth extraction and restoration |
||||
|
Pain (NRS) |
1.42 ± 2.54 |
1.69 ± 2.79 |
1.14 2.28 |
ns |
|
Pts with pain, n (%) |
22 (30.6) |
13 (36.1) |
9 (25.0) |
ns |
|
Intervention time (min) |
29 ± 13 |
30 ± 10 |
29 ± 16 |
ns |
|
Restoration |
||||
|
Pain (NRS scale) |
0.82 ± 1.90 |
0.53 ± 1.35 |
1.11 ± 2.33 |
ns |
|
Pts with pain, n (%) |
8 (21.1) |
3 (15.8) |
5 (26.3) |
ns |
|
Intervention time (min) |
27 ± 7 |
27 ± 6 |
0.26 ± 0.08 |
ns |
|
Extraction |
||||
|
Pain (NRS) |
2.09 ± 3.00 |
3.00 ± 3.39 |
1.18 ± 2.30 |
.058 |
|
Pts with pain (%) |
14 (41.2%) |
10 (58.8%) |
4 (23.5%) |
.037 |
|
Intervention time (min) |
32 ± 18 |
32 ± 13 |
32 ± 22 |
ns |
NRS= Numeric Rating Scale
Table 3: Primary Endpoint Results.
|
Arrhythmia/Study Intervals |
Total group N=72 |
Lidocaine without epinephrine N=36 |
Lidocaine with epinephrine N=36 |
p |
|
Ventricular extra-systoles |
ns |
|||
|
Baseline (for 1 hour) |
194 ± 403 |
264 ± 519 |
122 ± 214 |
ns |
|
Anesthesia (15 minutes) |
51 ± 104 |
71 ± 136 |
30 ± 50 |
ns |
|
Intervention |
91 ± 209 |
121 ± 242 |
63 ± 173 |
ns |
|
Post intervention (for 1 hour) |
95 + 182 |
126 ± 232 |
63 ± 101 |
ns |
|
Atrial extra-systoles |
||||
|
Baseline (for 1 hour) |
17 ± 36 |
15 ± 29 |
19 ± 41 |
ns |
|
Anesthesia (15 minutes) |
8 ± 14 |
11 ± 17 |
6 ± 13 |
ns |
|
Intervention |
13 ± 20 |
11 ± 18 |
15 + 23 |
ns |
|
Post intervention (for 1 hour) |
25 ± 45 |
19 ± 37 |
31 ± 52 |
ns |
|
No sustained V tachycardia* |
||||
|
Baseline (for 1 hour) |
36 ± 90 |
62 ± 119 |
2 ± 1 |
- |
|
Anesthesia (15 minutes) |
5 ± 3 |
8 |
3 |
- |
|
Intervention |
11 ± 15 |
20 ± 19 |
2 ± 1 |
- |
|
Post intervention (for 1 hour) |
5 ± 6 |
5 ± 7 |
3 ± 4 |
- |
|
Supraventricular tachycardia* |
||||
|
Baseline (for 1 hour) |
31 ± 50 |
31 ± 50 |
-- |
- |
|
Anesthesia (15 minutes) |
2 ± 2 |
2 ± 2 |
-- |
- |
|
Intervention |
29 ± 40 |
29 ± 40 |
-- |
- |
|
Post intervention (for 1 hour) |
16 ± 24 |
23 ± 30 |
4 ± 4 |
- |
V= ventricular; * number not enough for comparison
Table 4: Arrhythmia Results.
Figure 2: Boxplot showing the groups LCE (lidocaine plus epinephrine) and LSE (lidocaine without epinephrine); (A) mean heart rate; (B) mean systolic blood pressure and (C) mean diastolic blood pressure during the pre-intervention, anesthesia, dental intervention, and post dental intervention periods.
4. Discussion
To the best of our knowledge, this is a pioneering prospective, controlled, double-blinded, trial testing the superiority of anesthesia plus vasoconstrictor compared with anesthesia alone having pain as a primary endpoint with HF patients. Results from our trial have shown [1] poor oral health with a high DMFt index (Decayed, Missing and Filled teeth); periodontal disorders were observed in all heart failure patients; [2] vasoconstrictor added to local anesthesia was superior to anesthesia alone for pain control during tooth extraction; [3] patients who received vasoconstrictor added to anesthesia needed less anesthetic than patients who received anesthesia alone; [4] all patients treated with both kinds of anesthetics had increased blood pressure during and after dental procedures compared with baseline, but no difference occurred between groups; [5] both groups had lots of arrhythmias, but this was not influenced by the procedures; [6] although this is a high-risk population with a very high number of arrhythmias and low LVEF, no adverse cardiovascular events were observed in either group.
Our findings of only 15.3% of patients with good oral health are lower than data from the Australian Prospective 45 and Up Study with over 150 000 participants, and concordant with the higher prevalence of severe periodontitis among chronic heart failure patients [10, 11]. Our results indicate a high DMFt index with a high prevalence of gingivitis and periodontal disease. This finding has importance, considering Australian Prospective 45 and Up Study results that showed that in reporting no teeth versus ≥20 teeth left, risks were increased for HF and all-cause mortality. Also, incidence of cardiovascular hospitalization risk increased significantly with increasing tooth loss for all outcomes except ischemic stroke [12]. Although lacking in empirical scientific evidence, a theory has been proposed that chronic infections or inflammatory processes in the oral cavity may cause systemic diseases [13]. The proposed biological pathway linking periodontal diseases and cardiovascular disease is thought to be through a low-grade systemic chronic inflammatory response caused by oral infections or inflammatory processes [14, 15]. Oral health and CHF share risk factors, such as smoking, diabetes mellitus, alcohol consumption, hypertension, and low socioeconomic status [16]. Despite little published data on the association between heart failure and oral health, our results and the Australian Prospective 45 and Up Study data guarantee the development of well-designed studies to test the hypothesis of oral heath being a marker for HF or even having a causative relationship.
Our results showing a reduction in pain with 2% lidocaine plus epinephrine agrees with reported results of a double-blind, prospective, randomized study in which 59 valvular heart disease patients were included for tooth extraction and restorations [17]. However, in this valvular trial, pain reduction was not a primary endpoint. Also, reduction of pain during dental extraction in patients receiving anesthesia with vasoconstrictor compared with anesthesia alone was reported in a randomized, non double-blind study in patients with coronary artery disease [18]. However, in both studies, patients had higher left ventricular function, fewer arrhythmias, and lower risk of HF compared with our HF population. In both, pain reduction was not a primary endpoint. Other studies have reported pain during dental intervention but comparing types of anesthetic drugs [19].
Concerning safety of a vasoconstrictor plus local anesthesia, our results can only be partially compared with other trials, considering that we have not found studies with the same high-risk HF population, analyzing heart rate, arrhythmias, and hemodynamic changes with the same design and same doses of local anesthesia. Publications have not reported events in patients who underwent dental intervention receiving anesthesia with vasoconstrictor that had valve disease, cardiomyopathy, and ischemic heart disease [20]; or in patients with controlled hypertension, ischemic heart disease, and HF [21]; and no effect on cardiac arrhythmia status in the ambulatory geriatric population [22]. No difference has been reported between patients with coronary artery disease receiving anesthesia with or without vasoconstrictor during dental intervention [23]; no difference in patients within three weeks of uncomplicated acute myocardial infarction [24]; no difference in patients with Chagas' disease or coronary artery disease who had complex ventricular arrhythmia [25]; and no additional ischemic risks in patients with coronary artery disease [26]. However, our results disagree with a retrospective report of 22% incidence of circulatory complications during dental treatment in patients with hypertrophic cardiomyopathy, dilated cardiomyopathy, or dilated phase of hypertrophic cardiomyopathy [27]; or induced atrial or ventricular arrhythmias during dental implant surgeries in healthy patients [28].
4.1 Limitations
More complex dental interventions were not studied. However, restorations and tooth extractions are common procedures in dental practice. Extra cartridges for anesthesia with vasoconstrictor could be a confounder for arrhythmia, systemic blood pressure, and heart rate analysis. However, it would be impossible to continue dental procedures without pain control. This trial has not studied different doses of anesthetics, which is also a limitation.
4.2 Clinical implication
Because HF is a highly prevalent disease in the population with risk of cardiac events, the treatment and prevention of its high incidence of tooth decay and periodontal diseases may be a challenge for cardiologists and dentists in clinical practice. Many factors, such as fear of pain, pain with hemodynamic and arrhythmic consequences, anxiety, potential need for a greater dose of anesthesia, and safety concerns about vasoconstrictors may play a role in this scenario. Our results showing that association of a vasoconstrictor with the anesthetic is beneficial, without compromising safety of this high-risk population, might introduce new evidence for improvement in care and prevention for patients with poor oral health. Also, the hypothesis of poor oral health treatment leading to a reduction in HF events should be tested in future trials.
5. Conclusions
The association of a vasoconstrictor with local anesthetic has shown to be superior compared with anesthesia without a vasoconstrictor, with lower incidence of pain during more invasive dental procedures, and with less need for additional anesthesia. The association of a vasoconstrictor with a local anesthetic is safe for this high-risk population, not causing adverse hemodynamic and arrhythmic events in heart failure patients.
Clinical perspectives
The impairment of oral and dental health frequently found in patients with heart failure is a challenge for the cardiologist and dentist. A basic care plan for patients with heart failure should, at a minimum, correspond to recommended prevention strategies for healthy teeth. Our results showing that the association of a vasoconstrictor with the anesthetic is beneficial, without compromising the safety of this high-risk population, may introduce new evidence of improvement in care and prevention for patients with poor oral health.
Translational Outlook
The impact of poor oral health seems to be associated with cardiovascular diseases. The treatment and prevention of its high incidence of dental caries and periodontal diseases can be a challenge for cardiologists and dentists in clinical practice. The results of the Tooth-HF Study translate and improve clinical and dental care in patients with heart failure.
References
- Mattila KJ, Nieminen MS, Valtonen VV, Rasi VP, Kesäniemi YA, et al. Association between dental health and acute myocardial infarction. BMJ 298 (1989): 779-781.
- Fröhlich H, Herrmann K, Franke J, Karimi A, Täger T, et al. Periodontitis in chronic heart failure. Tex Heart Inst J43 (2016): 297-304.
- Holmlund A, Holm G, Lind L. Number of teeth as a predictor of cardiovascular mortality in a cohort of 7, 674 subjects followed for 12 years. J Periodontol 81(2010): 870-876.
- Bahekar AA, Singh S, Saha S, Molnar J, Arora R. The prevalence and incidence of coronary heart disease is significantly increased in periodontitis: a meta-analysis. Am Heart J 154 (2007): 830-837.
- Berent R, Auer J, Schmid P, Krennmair G, Crouse SF, et al. Periodontal and coronary heart disease in patients undergoing coronary angiography. Met C Exp 60 (2011): 127-133.
- Reynolds MA. Modifiable risk factors in periodontitis: at the intersection of aging and disease. Periodontol 2000 64 (2014): 7-19.
- Benjamin EJ, Virani SS, Callaway CW, Chamberlain AM, Chang AR, et al. American Heart Association Council on Epidemiology and Prevention Statistics Committee and Stroke Statistics Subcommittee. Heart disease and stroke statistics-2018 update: a report from the American Heart Association. Circulation 137 (2018): e67-e492.
- Devereaux PJ, Chan MT, Alonso-Coello P, Walsh M, Berwanger O, et al. Vascular events in noncardiac surgery patients cohort evaluation (VISION) study investigators. Association between postoperative troponin levels and 30-day mortality among patients undergoing noncardiac surgery. JAMA 307 (2012): 2295-2304.
- Laragnoit AB, Neves RS, Neves IL, Vieira JE. Locoregional anesthesia for dental treatment in cardiac patients: a comparative study of 2% plain lidocaine and 2% lidocaine with epinephrine (1:100, 000). Clinics (Sao Paulo) 64 (2009): 177-182.
- Kamath M, Mala K, Thomas MS. Modification of Dental Care for Patients with Cardiac Disease. OHDM 15 (2016): 286-290.
- Joshy G, Arora M, Korda RJ, Chalmers J, Banks E. Is poor oral health a risk marker for incident cardiovascular disease hospitalisation and all-cause mortality? Findings from 172 630 participants from the prospective 45 and Up Study. BMJ Open 6 (2016): e012386.
- Watt RG, Tsakos G, de Oliveira C, Hamer M. Tooth loss and cardiovascular disease mortality risk--results from the Scottish Health Survey. PLoS One 7 (2012): e30797.
- Lockhart PB, Bolger AF, Papapanou PN, Osinbowale O, Trevisan M, et al. Periodontal disease and atherosclerotic vascular disease: does the evidence support an independent association?: a scientific statement from the American Heart Association. Circulation 125 (2012): 2520-2544.
- Niwa H, Sato Y, Matsuara M. Safety of dental treatment in patients with previously diagnosed acute myocardial infarction or unstable angina pectoris. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 89 (2000): 35-41.
- Hupp JR. Ischemic Heart Disease: Dental Management Considerations. Dent Clin N Am 50 (2006): 483-491.
- Reynolds MA. Modifiable risk factors in periodontitis: at the intersection of aging and disease. Periodontol 2000 64 (2014): 7-19.
- Laragnoit AB, Neves RS, Neves IL, Vieira JE. Locoregional anesthesia for dental treatment in cardiac patients: a comparative study of 2% plainlidocaine and 2% lidocaine with epinephrine (1:100, 000). Clinics; 64 (2009):177-182.
- Conrado VC, de Andrade J, Timerman L, Andrade MM, Moreira DR, et al. Cardiovascular effects of local anesthesia with vasoconstrictor during dental extraction in coronary patients. Arq Bras Cardiol 88 (2007): 507-513.
- Elad S, Admon D, Kedmi M, Naveh E, Benzki E, et al. The cardiovascular effect of local anesthesia with articaine plus 1:200, 000 adrenaline versus lidocaine plus 1:100, 000 adrenaline in medically compromised cardiac patients: a prospective, randomized, double blinded study. Oral Surg Oral Med Oral Path Oral Radiol Endod 105 (2008): 725-730.
- Niwa H, Sugimura M, Satoh Y, Tanimoto A. Cardiovascular response to epinephrine-containing local anesthesia in patients with cardiovascular disease. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 92 (2001): 610-616.
- Laragnoit AB, Neves RS, Neves, ILI, Vieira JE. Locoregional anesthesia for dental treatment in cardiac patients: a comparative study of 2% plain lidocaine and 2% lidocaine with epinephrine (1:100, 000). Clinics 64 (2009): 177-182.
- Campbell JH, Huizinga PJ, Das SK, Rodriguez JP, Gobetti JP. Incidence and significance of cardiac arrhythmia in geriatric oral surgery patients. Oral Surg Oral Med Oral Pathol Oral Radiol Endod 82 (1996): 42-46.
- Neves RS, Neves IL, Giorgi DM, Grupi CJ, César LA, et al. Grinberg M Effects of epinephrine in local dental anesthesia in patients with coronary artery disease. Arq Bras Cardiol 88 (2007): 545-551.
- Cintron G, Medina R, Reyes AA, Lyman G. Cardiovascular effects and safety of dental anesthesia and dental interventions in patients with recent uncomplicated myocardial infarction. Arch Intern Med 146 (1986): 2203-2204.
- Cáceres MT, Ludovice AC, Brito FS, Darrieux FC, Neves RS, et al. Effect of local anesthetics with and without vasoconstrictor agent in patients with ventricular arrhythmias. Arq Bras Cardiol91 (2008): 128-133.
- Conrado VC, de Andrade J, de Angelis GA, de Andrade AC, Timerman L, et al. Cardiovascular effects of local anesthesia with vasoconstrictor during dental extraction in coronary patients. Arq Bras Cardiol 88 (2007): 507-513.
- Shibuya M, Kamekura N, Kimura Y, Fujisawa T, Fukushima K. Clinical study of anesthetic management during dental treatment of 25 patients with cardiomyopathy. Spec Care Dentist ;23 (2003): 216-222.
- Romano MM, Soares MS, Pastore CA, de Oliveira Guaré R, Adde CA. Electrocardiographic alterations during endosseous implant placement performed with local anesthetic agents. Int J Oral Maxillofac Implants 24 (2009): 412-418.